Are ionic interactions permanent?
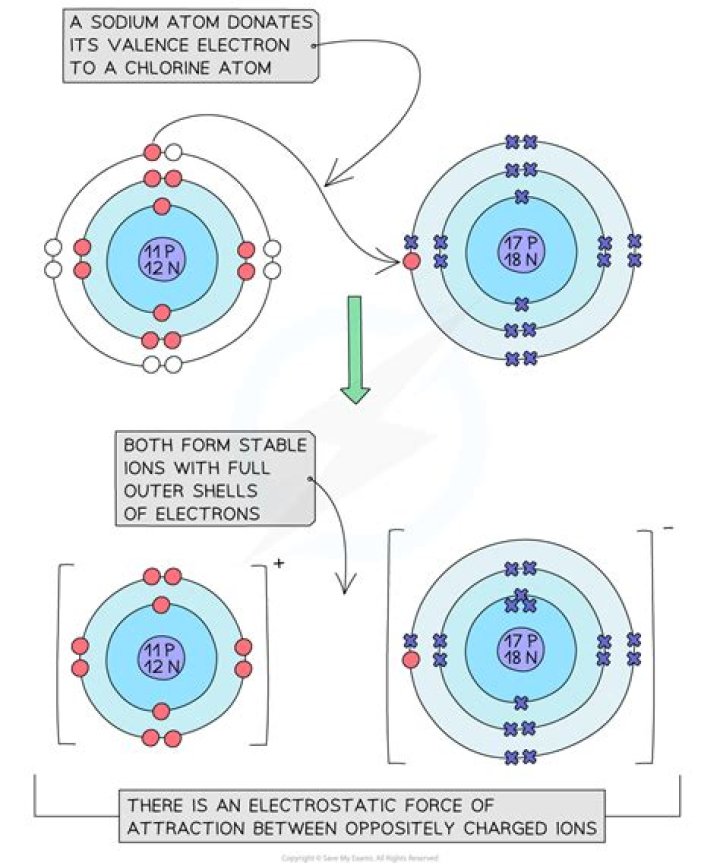
Ionic interactions involve the attraction of ions or molecules with full permanent charges of opposite signs.
What are electrostatic interactions?
1 Electrostatic interactions. Electrostatic interactions comprise the attractive or repulsive interactions between charged molecules. In most cases, electrostatic interactions are generally combined with other interactions for material repair.
What is a permanent dipole?
As we have seen in the polarity section, a permanent dipole is created in a molecule when there is an electronegative atom, such as a oxygen, nitrogen or a halogen. The permanent dipole consists of regions of partial positive charge and regions of partial negative charge within the same molecule.
Are dispersion forces permanent or temporary?
The London dispersion force is a temporary attractive force that results when the electrons in two adjacent atoms occupy positions that make the atoms form temporary dipoles. This force is sometimes called an induced dipole-induced dipole attraction.
Are electrostatic interactions covalent?
electrostatic interactions Definition The intermolecular forces of attraction in which complete or partial ionic species are attracted to each other are termed as electrostatic interactions. These attraction forces do not include any sharing of electrons between atoms. So, they are also named as non-covalent bonds.
Are electrostatic interactions the same as ionic bonds?
Electrostatic interactions are also known as Charge-charge interactions and Ionic interactions. An electrostatic attraction exists when there are closely packed ions of opposite charges. An electrostatic repulsion is present between different ions that have the same charge.
Are electrostatic interactions ionic interactions?
What are electrostatic and bonding interactions?
Electrostatic interaction (van der Waals interaction): The attractive or repulsive interaction between objects having electric charges. Electrostatic attraction (shown in red) between the δ+ and δ- ends of a polar covalent N-H bond allow for hydrogen bonding and base pairing within the DNA double helix.
Are induced dipoles permanent?
Dipoles. There are two kinds of dipole moments: Permanent electric dipole moments can arise when bonding occurs between elements of differing electronegativities. Induced (temporary) dipole moments are created when an external electric field distorts the electron cloud of a neutral molecule.
What molecules have permanent dipoles?
Molecules that contain dipoles are called polar molecules and are very abundant in nature. For example, a water molecule (H2O) has a large permanent electric dipole moment.
Which intermolecular force does not require a permanent dipole?
dispersion forces
Therefore dispersion forces and dipole-dipole forces act between pairs of PF3 molecules. (c) CO2 is a linear molecule; it does not have a permanent dipole moment; it does contain O, however the oxygen is not bonded to a hydrogen. Therefore only dispersion forces act between pairs of CO2 molecules.
What are electelectrostatic interactions?
Electrostatic interactions are a type of attraction forces in which complete or partially ionic species are attracted to each other. Further, this term includes both attractive and repulsive forces between ionic species, i.e. oppositely charged ions are attracted to each other while the same charges repel from each other.
What are electrostatic interactions in materials repair?
Electrostatic interactions comprise the attractive or repulsive interactions between charged molecules. In most cases, electrostatic interactions are generally combined with other interactions for material repair. Although materials with the self-healing process based solely on electrostatic interactions are rare, such materials do exist.
What is the electrostatic interaction between ionic species?
Electrostatic interaction between ionic species of the opposite charges is a relatively strong interaction and often exists in solutions of highly polar solvents, especially aqueous solvents. Therefore, it can be used in the fluorescent sensing of chiral ions in aqueous environment.
What is the electrostatic interaction between the screened charges?
The electrostatic interaction between the screened charges is then short ranged. The original distribution is recovered by adding a second Gaussian charge distribution identical to the first, but of opposite sign.



