Can C form a triple bond?
Carbon-Carbon Bonds Carbon can form single, double, or even triple bonds with other carbon atoms. In a double bond, they share two pairs of electrons, and in a triple bond they share three pairs of electrons.
What is C triple bond called?
Alkynes are organic molecules made of the functional group carbon-carbon triple bonds and are written in the empirical formula of CnH2n−2. They are unsaturated hydrocarbons. Like alkenes have the suffix –ene, alkynes use the ending –yne; this suffix is used when there is only one alkyne in the molecule.
What is C triple bond C?
In organic chemistry, an alkyne is an unsaturated hydrocarbon containing at least one carbon—carbon triple bond. Alkynes are traditionally known as acetylenes, although the name acetylene also refers specifically to C 2H 2, known formally as ethyne using IUPAC nomenclature.
What type of bond is C?
covalent bond
5.1. 2 Carbon Bonding. A carbon–carbon bond is a covalent bond between two C atoms. The most familiar form is the single bond comprised of two electrons, one from each of the two atoms.
Is C double bond C polar?
When carbon forms multiple bonds to other elements, these bonds are polar. Both the carbon-oxygen double bond in formaldehyde (methanal) and the carbon—nitrogen triple bond in acetonitrile (cyanomethane) are polar….Polar Covalent Bonds.
| Structural Unit1 | Bond Moments (D) |
|---|---|
| C—Br | 1.4 |
| C—I | 1.2 |
| C = O | 2.3 |
| C ≡ N | 3.5 |
What is the bond length in C-C bond?
Bond strengths and lengths
| Molecule | Ethane | Acetylene |
|---|---|---|
| Structure | ||
| Hybridisation of carbon | sp3 | sp |
| C-C bond length | 1.535 Å | 1.203 Å |
| Proportion of C-C single bond | 100% | 78% |
What is an alkyne in chemistry?
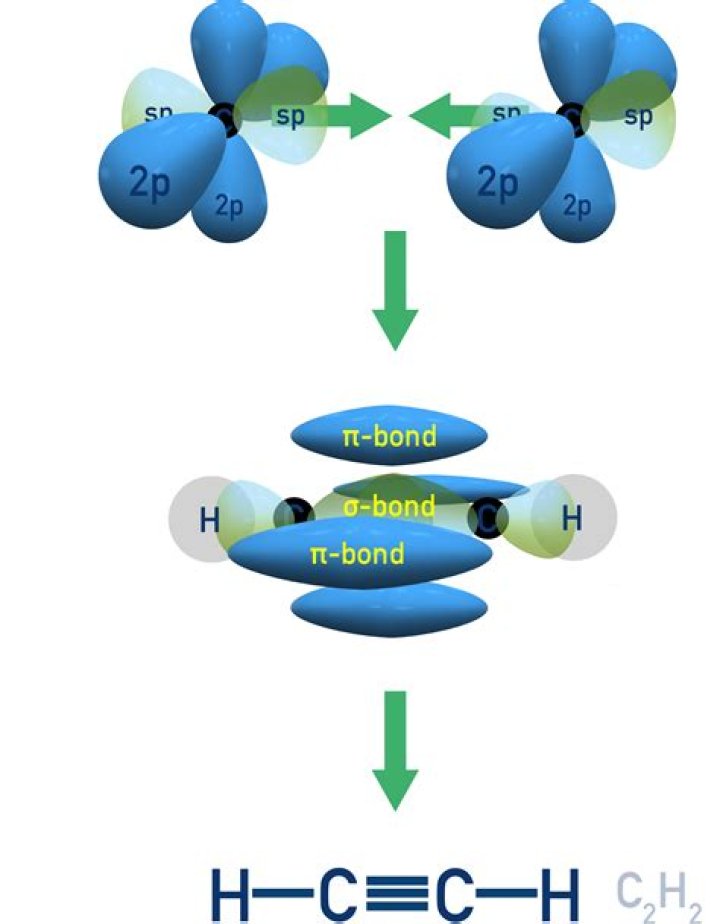
In chemical compound: Alkynes. Molecules that contain a triple bond between two carbon atoms are known as alkynes. The triple bond is made up of one σ bond and two π bonds.
What is hybridization of HCN?
In HCN, there are two sigma bonds, C-H and C-N. The number of sigma bonds is equal to the number of hybrid orbitals formed. So the hybridization for HCN molecule is sp hybridization.
Is C and O polar or nonpolar?
Single or multiple bonds between carbon atoms are nonpolar. Hydrogen and carbon have similar electronegativity values, so the C—H bond is not normally considered a polar covalent bond….Polar Covalent Bonds.
| Structural Unit1 | Bond Moments (D) |
|---|---|
| C—O | 0.7 |
| C—F | 1.4 |
| C—Cl | 1.5 |
| C—Br | 1.4 |
Is C-C polar or nonpolar?
Electronegativity and Bond Type
| Bond | Electronegativity Difference | Polarity |
|---|---|---|
| C–C | 0.0 | nonpolar |
| C–H | 0.4 | δ−C−δ+H |
| Si–C | 0.7 | δ+Si−δ−C |
| Si–O | 1.7 | δ+Si−δ−O |
What is C double bond C?
What is a Double Bond. A double bond is formed by sharing two pairs of valence electrons located in the outermost orbit of atoms. Compounds with double bonds are highly reactive than single bonded compounds but less reactive than compounds with triple bonds. Double bonds are denoted by two parallel dashes; ex: C=C.
What is the definition of triple covalent bond?
triple bond(Noun) A covalent bond in which three electron pairs (instead of the usual one) are shared between two atoms; most common between carbon atoms and carbon or nitrogen atoms, but a few other forms are known. Symbolized in formulae as u2261.
The carbon-hydrogen bond (C–H bond) is a bond between carbon and hydrogen atoms that can be found in many organic compounds. This bond is a covalent bond meaning that carbon shares its outer valence electrons with up to four hydrogens. This completes both of their outer shells making them stable.



