Can hydrogen be produced from biomass?
Biomass gasification is a mature technology pathway that uses a controlled process involving heat, steam, and oxygen to convert biomass to hydrogen and other products, without combustion.
How do you make hydrogen from biomass?
One method of generating hydrogen from biomass begins with processing the organic materials at high temperatures, but without combustion. When combined with oxygen and steam, the reaction results in carbon monoxide, carbon dioxide and hydrogen gas.
Can you make hydrogen from propane?
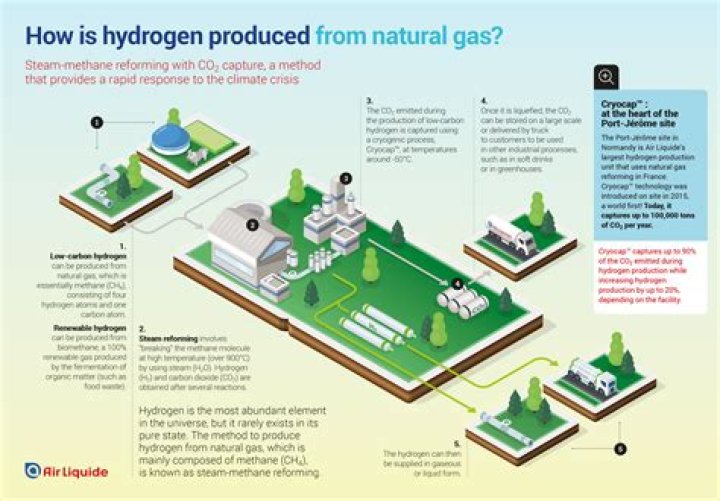
Hydrogen Production Over 95% of the world’s hydrogen is produced using steam methane reformers (SMR). Like methane, propane can be steam reformed, albeit at milder temperatures. The methane SMR reaction takes place at temperatures of 750°C to 950°C, but propane can be reformed to hydrogen at 200°C to 350°C.
What is the most efficient way to produce hydrogen?
The carbon monoxide is reacted with water to produce additional hydrogen. This method is the cheapest, most efficient, and most common. Natural gas reforming using steam accounts for the majority of hydrogen produced in the United States annually.
What are the products of gasification?
The main product of gasification is a mixture of gases (“producer gas”) with the main components H2, CO, CO2, H2O, CH4, and air nitrogen. This technology produces a gas with a low heating value (4–6 MJ/m3) and an 8–14 vol% H2 content only.
How does electrolysis produce hydrogen?
Electrolysis is a promising option for carbon-free hydrogen production from renewable and nuclear resources. Electrolysis is the process of using electricity to split water into hydrogen and oxygen. This reaction takes place in a unit called an electrolyzer.
How efficient is making hydrogen?
Very roughly, a new electrolysis plant today delivers energy efficiency of around 80%. That is, the energy value of the hydrogen produced is about 80% of the electricity used to split the water molecule. Steam reforming is around 65% efficient.
What products does gasification produce?
Gasification — known also as pyrolitic distillation — is a thermo-chemical process that converts biomass into a combustible gas called producer gas (syngas). Producer gas contains carbon monoxide, hydrogen, water vapor, carbon dioxide, tar vapor, and ash particles.
Is it possible to produce hydrogen from biomass gasification?
hydrogen is predominantly produced from fossil fuels. Hydrogen production from biomass via gasification can be an auspicious alternative for future decarbonized applications, which are based on renewable and carbon-dioxide-neutral produced hydrogen. This study gives an overview of possible ways to produce hydrogen via biomass gasification. First,
Is biomass gasification the future of biofuels?
Gasification plants for biofuels are being built and operated, and can provide best practices and lessons learned for hydrogen production. The U.S. Department of Energy anticipates that biomass gasification could be deployed in the near-term timeframe. What Is Biomass?
Why the yield of hydrogen is low from biomass?
The yield of hydrogen is low from biomass since the hydrogen content in biomass is low to begin with (approximately 6% versus 25% for methane) and the energy content is low due to the 40% oxygen content of biomass.
How do you separate hydrogen from a gas stream?
Adsorbers or special membranes can separate the hydrogen from this gas stream. Note: The above reaction uses glucose as a surrogate for cellulose. Actual biomass has highly variable composition and complexity with cellulose as one major component. Pyrolysis is the gasification of biomass in the absence of oxygen.



