Does NIH require DSMB?
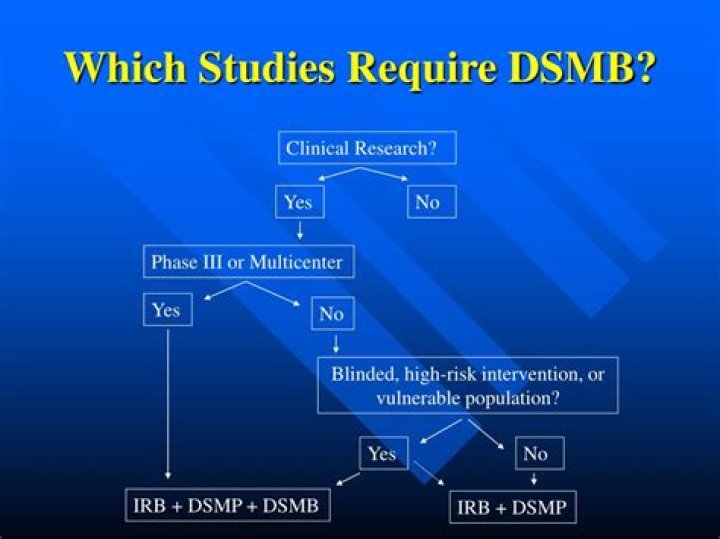
The NIH requires data and safety monitoring for all clinical trials. Multi-site clinical trials and most phase III clinical trials will require monitoring in the form of DSMBs. In June 2000, the NIH issued further guidance on data and safety monitoring for phase I and phase II trials.
What is a DSMB in clinical trials?
A Data and Safety Monitoring Board (DSMB) is a committee of experts responsible for reviewing clinical trial data on an ongoing basis to ensure the safety of study subjects and validity and integrity of the data. Members should be independent, with no vested interest in a specific treatment.
Who can be on a DSMB?
In general, the DSMB voting members are appointed by the clinical trial sponsor or by the Principal Investigator. A DMSB may have as few as three voting members; however, the number of members and the specific composition of the Board will depend on the type and complexity of the clinical trial.
Are DSMB members paid?
DSMB/OSMB members are paid an honorarium by the NIAMS for their participation in DSMB/OSMB meetings, which includes contributing their time and efforts to the review of the study materials and the ongoing review of safety reports, as well as other requests as needed.
How does a DSMB work?
The primary responsibilities of the DSMB are to 1) periodically review and evaluate the accumulated study data for participant safety, study conduct and progress, and, when appropriate, efficacy, and 2) make recommendations to NIDCR concerning the continuation, modification, or termination of the trial.
What is a DSMB NIH?
The Data and Safety Monitoring Board (DSMB) is an independent group of experts that advises NIDCR. During the trial, the DSMB should review cumulative study data to evaluate safety, study conduct, and scientific validity and integrity of the trial.
What is DSMB report?
A Data and Safety Monitoring Board (DSMB) is an independent group of experts that objectively evaluates trial data periodically for integrity and to assure the safety of clinical trial participants.
How often should DSMB meetings be held?
once annually
Once a study is implemented, the DSMB should convene as often as necessary, but at least once annually, to examine the accumulated safety and enrollment data, review study progress, and discuss other factors (internal or external to the study) that might impact continuation of the study as designed.
How much do DSMB members get paid?
DSMB members or their family members should have no professional, proprietary or financial relationship with the sponsoring companies, and the NIAID DSMB executive secretary vetted all members for potential conflicts of interest, NIAID said in response to questions from KHN. Members are paid $200 per meeting.
Is DSMB the same as IRB?
Clinical trials have two independent review bodies that make participant safety a number one concern as they plan, implement and monitor trial activities: an Institutional Review Board (IRB) and a Data Safety Monitoring Board (DSMB). These plans are also referred to as a protocol.
What is Susar in pharmacovigilance?
SAR is the abbreviation for Serious Adverse Reaction, and SUSAR for Suspected Unexpected Serious Adverse Reaction.
What types of clinical trials require DSMBs?
Multi-site clinical trials and most phase III clinical trials will require monitoring in the form of DSMBs. In June 2000, the NIH issued further guidance on data and safety monitoring for phase I and phase II trials.
What is a DSMB and why is it important?
The purpose of the DSMB is to provide oversight and monitoring of the conduct of clinical trials to ensure the safety of participants and the validity and integrity of study data. The National Institutes of Health (NIH) strongly recommends data and safety monitoring in the form of a DSMB for all Phase III clinical trials.
What are the NIAMS DSM guidelines for clinical research?
These guidelines replace the NIAMS Policy for Data and Safety Monitoring of Clinical Research released on August 1, 2017. The NIAMS DSM guidelines apply to all NIAMS-funded grants, cooperative agreements, or contracts that include clinical research.
What is the NIMH data and safety monitoring guidance for clinical trials?
This guidance applies to data and safety monitoring for all NIMH-supported clinical trials (including grants, cooperative agreements, and contracts). It is the policy of the National Institutes of Health (NIH) that each Institute and Center (I/C) have a system in place for the appropriate oversight and monitoring of NIH-funded clinical trials.



