How are amines named?
Amines are named by naming the alkyl groups attached to the nitrogen atom, followed by the suffix -amine.
What is the formula of aromatic amines?
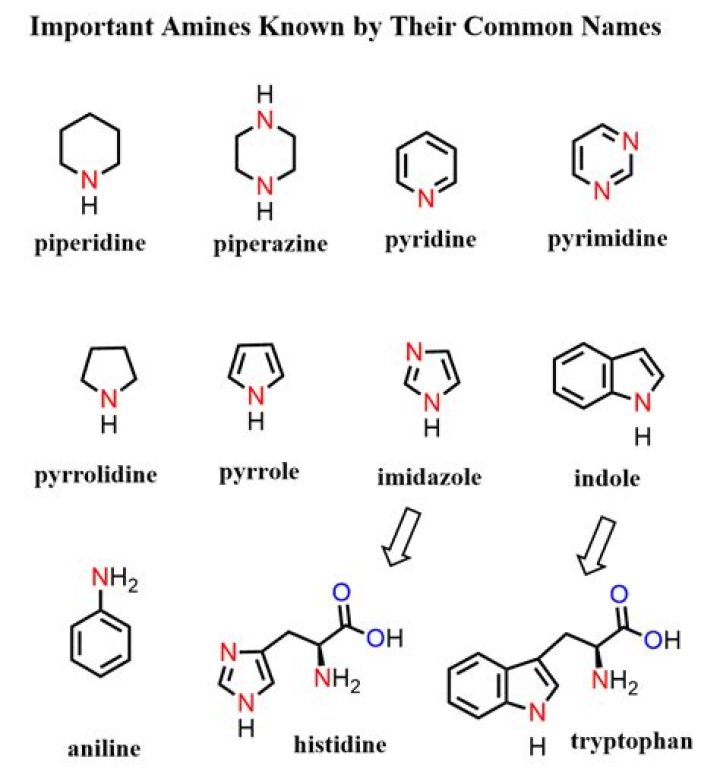
The simplest aromatic amine CH6H5NH2 is called benzenamine ( Aniline). Other aromatic amines are named as derivatives of benzenamine, and the positions of other groups are indicated by numbers. Q.
What is the common name of the simplest aromatic amine?
What is the common name of the simplest aromatic amine? Explanation: The simplest amine is C6H5NH2 and is known as aniline. This name is also accepted as the IUPAC name. Its actual IUPAC name is benzenamine, or it may also be written as aminobenzene.
What is the name of the simplest aromatic amine?
Aniline (benzenamine) is the simplest of the primary aromatic amines. Aromatic amines can be produced by reduction of the corresponding nitro compound, the ammonolysis of an aromatic halide or phenol, and by direct amination of the aromatic ring.
What is an example of an aromatic hydrocarbon?
Some examples of aromatic hydrocarbons are Methylbenzene, Naphthalene, Phenanthrene, Trinitrotoluene, and o-dihydroxybenzene.
What is the nomenclature for amines?
The Chemical Abstract Service has adopted a nomenclature system in which the suffix -amine is attached to the root alkyl name. For 1º-amines such as butanamine (first example) this is analogous to IUPAC alcohol nomenclature (-ol suffix). The additional nitrogen substituents in 2º and 3º-amines are designated by the prefix N- before the group name.
What is the difference between aliphatic and aromatic amine?
Aliphatic amine: a nitrogen is bonded only to alkyl groups. Aromatic amine: a nitrogen is bonded to one or more aryl groups ( an aryl group – a functional group derived from an aromatic ring ). Heterocyclic amine: an amine in which nitrogen is one of the atoms of a ring.
How do you find the amine group number?
The amine group is located by the position number. Groups that are attached to the nitrogen atom are located using “N” as the position number. More complex primary amines are named with —NH2 as the amino substituent. • Aromatic amines: named as derivatives of the parent compound aniline.
What is the difference between an alcohol and an amine?
Note the difference in classification of alcohols and amines: Alcohols are classified by the number of alkyl groups on the a- carbon, but amines (as is with amides) are classified by the number of alkyl or aryl groups attached to the nitrogen.



