How are ylides stabilized?
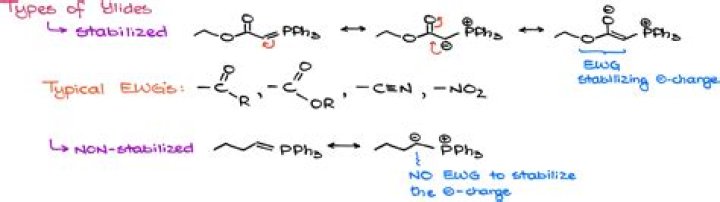
ii) Stabilized ylides: The ylides with electron withdrawing groups adjacent to the negatively charged carbon are more stable. These are usually stabilized by conjugation. * It is generally observed that the geometry of the final alkene depends on the stability of the ylide.
What are Sulphur Ylides give an example?
The sulphur ylide is usually generated by deprotonation of a sulphonium salt with a base. For example, the treatment of a sulphonium salt 9.49 with NaOMe gives the product via a [2,3] sigmatropic rearrangement (Fig. 9.62).
Which is most important Sulphur Ylides?
The most important sulphur ylides are dimethylsulfonium methylide and dimethylsulfoxonium methylide (Corey-Chaykovsky reagent). These ylides are prepared by deprotonation of the corresponding sulfonium salts, which can be prepared from the reaction of either dimethylsulphide or dimethylsulphoxide with methyl iodide.
What reacts with ylides?
Organophosphorus ylides react with aldehydes or ketones to give substituted alkenes in a transformation called the Wittig reaction.
Which ketone is slower in Wittig reaction?
A few limitations of the Wittig reaction are: Both the E and the Z double bond isomers can be formed. The reaction speed is very slow when sterically hindered ketones are used.
Which of the following ylides are least stable?
Sigmatropic rearrangements. Many ylides react in sigmatropic reactions. The Sommelet-Hauser rearrangement is an example of a [2,3]-sigmatropic reaction. The Stevens rearrangement is a [1,2]-rearrangement.
Which of the following base is commonly used for the generation of phosphorus Ylides?
Sodium hydride (NaH), another powerful base, is insoluble in most solvents, but its reaction with DMSO (the weakest acid in the table) generates a strong conjugate base, CH3)S(=O)CH2(–) Na(+), known as dimsyl sodium. This soluble base is widely used for the generation of ylides in DMSO solution.
How Enamines are formed give the reaction?
Enamines are formed by the reaction of secondary amines with carbonyl compounds. Common secondary amines used to form enamines include pyrrolidine, piperidine, and morpholine. Enamines react as nucleophiles, resulting in alkylation at the position equivalent to the a carbon atom of the original carbonyl compound.
What does the Wittig reaction do?
The Wittig Reaction allows the preparation of an alkene by the reaction of an aldehyde or ketone with the ylide generated from a phosphonium salt.
Which one of the aldehyde is slowly reacted in Wittig reaction?
The Wittig reaction or Wittig olefination is a chemical reaction of an aldehyde or ketone with a triphenyl phosphonium ylide called a Wittig reagent….
| Wittig reaction | |
|---|---|
| aldehyde or ketone + triphenyl phosphonium ylide ↓ alkene + triphenylphosphine oxide | |
| Conditions | |
| Typical solvents | typically THF or diethyl ether |
| Identifiers |



