How can we identify the products from baking soda and calcium chloride reaction?
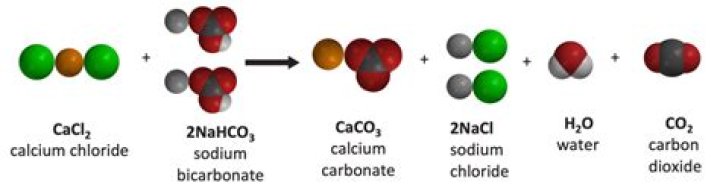
Combine sodium bicarbonate (baking soda), calcium chloride and water and you get calcium carbonate (a chalky precipitate) plus carbon dioxide gas, sodium chloride (table salt), hydrochloric acid and a fair amount of heat.
What happens if you mix salt and baking soda?
Description: This video shows a chemical reaction of sodium bicarbonate (baking soda), calcium chloride (road salt), and an indicator phenol red. The chemicals react to form calcium carbonate, sodium chloride, and carbon dioxide gas.
What occurs when baking soda and calcium chloride are combined?
Calcium chloride, baking soda, and water combine to produce carbon dioxide gas. Phenol red is an acid–base indicator that changes color in the presence of acids and bases.
What kind of change physical or chemical occurred when CaCl2 and nahco3 were combined?
This is a chemical change because new products were formed as a result of this reaction. We learned that phenol red turns yellow in an acid. When Calcium chloride and Sodium bicarbonate are mixed with water, the CO2 gas that is produced will react with the water and make the water acidic (carbonic acid).
What happens when you mix baking soda and calcium chloride?
Is calcium carbonate baking soda?
Baking soda comes in two forms: sodium bicarbonate and calcium carbonate.
What happens when you mix calcium chloride and baking soda?
What is the reaction between sodium carbonate and calcium chloride?
The reaction is: Na2CO3 (aq) + CaCl2(aq) → CaCO3 (s) + 2 NaCl (aq) We will use approximately 0.02 mole of each reactant and expect to obtain approximately 0.02 mole of solid product, since the stoichiometric coefficients are all 1 in the balanced equation.
Is calcium chloride and baking soda a chemical or physical change?
The dissolving of calcium chloride and baking soda in water are physical changes even though they release or absorb energy. (The solids could be reclaimed if the water were evaporated.) The formation of calcium carbonate, carbon dioxide and carbonic acid are chemical changes.
What happens when you combine calcium chloride & baking soda?
Combining calcium chloride and baking soda — sodium bicarbonate — in a sealable plastic bag is a favorite high school chemistry experiment. It produces a gas, so if you seal the bag after combining the chemicals, the bag will blow up like a balloon.
How do you make calcium carbonate from baking soda and water?
Combine sodium bicarbonate (baking soda), calcium chloride and water and you get calcium carbonate (a chalky precipitate) plus carbon dioxide gas, sodium chloride (table salt), hydrochloric acid and a fair amount of heat.
How do you make phenol red with calcium chloride and baking soda?
11 g of calcium chloride. 10 ml phenol red solution. Small vial for phenol red solution. Phenol red solution is made by adding four drops of phenol red to 10 ml of distilled water. Measure out the baking soda and calcium chloride prior to the demo and put each in a separate beaker.
How do you use vinegar and baking soda to test for temperature?
Pour about 10 mL of vinegar into a small plastic cup. Then, place a thermometer into the vinegar. Record the initial temperature (T i) in the table below. While the thermometer is in the cup, add about ½ teaspoon of baking soda to the cup. Watch the thermometer for any change in temperature.



