How do cyclohexane and benzene differ?
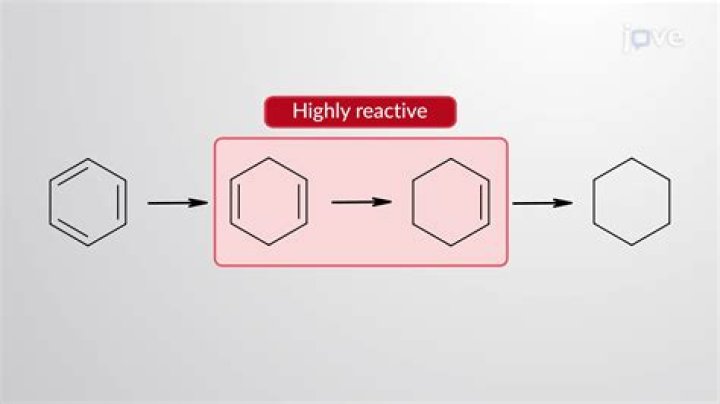
The key difference between benzene and cyclohexane is that benzene is an aromatic compound whereas cyclohexane is a non-aromatic compound. It is because, there are no double bonds between carbon atoms in the cyclohexane ring.
What is the difference between benzene and benzene?
Benzene and benzine are not the same thing. Though they are often confused or used interchangeably in books and magazines, they are very different. Benzene is spelled with an “e” as in dead. Benzine is spelled with an “i” as in alive.
Are cyclohexane and benzene isomers?
In other words, cyclohexane is not the same as benzene! These two compounds have different molecular formulas and their chemical and physical properties are not the same. The hydrogenation technique can be used by chemists to convert from benzene to cyclohexane by saturating the benzene ring with missing hydrogens.
What is the structure of cyclohexane and benzene?
Benzene (C6H6) has six C-atoms and six H-atoms, it contains three double bonds alternately between two C-atoms. Cyclohexane (C6H12) has six C-atoms each possessing two H-atoms, thus twelve H-atoms total. It does not consist any double bond.
Is benzene more stable than cyclohexane?
If it is benzene, then how come the heat of hydrogenation of benzene to cyclohexane is an exothermic process which releases energy, indicating that cyclohexane is more stable.
What is the difference between cyclohexane and hexane?
The key difference between hexane and cyclohexane is that hexane is an acyclic alkane while cyclohexane is a cyclic alkane with a ring structure. They both have six carbon atoms, but a different number of hydrogen atoms.
Is cyclohexane an aromatic compound?
Aromaticity. Cyclic compounds may or may not exhibit aromaticity; benzene is an example of an aromatic cyclic compound, while cyclohexane is non-aromatic.
Why is benzene called benzene?
The word “benzene” derives from “gum benzoin” (benzoin resin), an aromatic resin known to European pharmacists and perfumers since the 16th century as a product of southwest Asia. An acidic material was derived from benzoin by sublimation, and named “flowers of benzoin”, or benzoic acid.
Is cyclohexane safer than benzene?
Cyclohexane, with the chemical formula C 6 H 12 , is a volatile, colorless, flammable, water-insoluble, non-corrosive, non-polar liquid having a pungent odor. It is less toxic than benzene. The most stable chemical structure of cyclohexane, known as ‘chair conformation’, is shown in Fig.
Why is cyclohexane weaker than benzene?
Benzene is less reactive with electrophiles than cyclohexene because the delocalised pi system has a lower electron density than the localised pi bond in the C=C double bond. This also means benzene cannot polarise bonds to generate nucleophiles, so reactions may need to take place in the presence of a halogen carrier.
Which is more stable cyclohexene or cyclohexane?
Another difference between cyclohexane and cyclohexene is that the cyclohexane is relatively stable, thus, is less reactive while the cyclohexene is relatively unstable, thus, can undergo reactions due to the presence of a double bond in the ring structure.
Why is cyclohexane used instead of hexane?
This is due to the fact that cyclohexane has 2 hydrogens less compared to hexane. The volume cyclohexane occupies is lower than hexane due to its cyclic structure. Hence, there’s a higher number of cyclohexane molecules in a given molecule when compared with hexane.
What is the difference between cyclohexane and benzene and cycloalkane?
It is a cycloalkane consisting of six single bonds and no multiple bonds. Cyclohexane,according to Baeyer’s strain theory,exhibits conformational isomerism. It exists mostly in the stable chair form . Other possible conformations are boat,twist boat and half chair. Benzene is an aromatic compound consisting of three single and three double bonds.
What is the chemical name of cyclohexane?
Cyclohexane is a cyclic molecule with the formula of C 6 H 12. Though it has a similar number of carbons like benzene, cyclohexane is a saturated molecule. Hence, there are no double bonds between carbons as in benzene. Also, it is a colourless liquid with a mild sweet odour.
What is the hybridization of the carbon atoms in cycyclohexane?
Cyclohexane is an organic compound having the chemical formula C 6H 12 and is a cyclic structure. It is a cycloalkane, which means, cyclohexane is a saturated compound having no double or triple bonds in its structure and is a cyclic compound. Therefore, all carbon atoms in cyclohexane are sp 3 hybridized.
What is the aromatic circle of cyclohexane?
Cyclohexane does not have an aromatic circle. Explanation: Cyclohexane has no double bonds between the carbons, its formula is C6H12, while benzene has 3 double bonds with the formula C6H6. Those double bonds are conjugated with the rest of the molecule, which is what we call an “aromatic” cycle.



