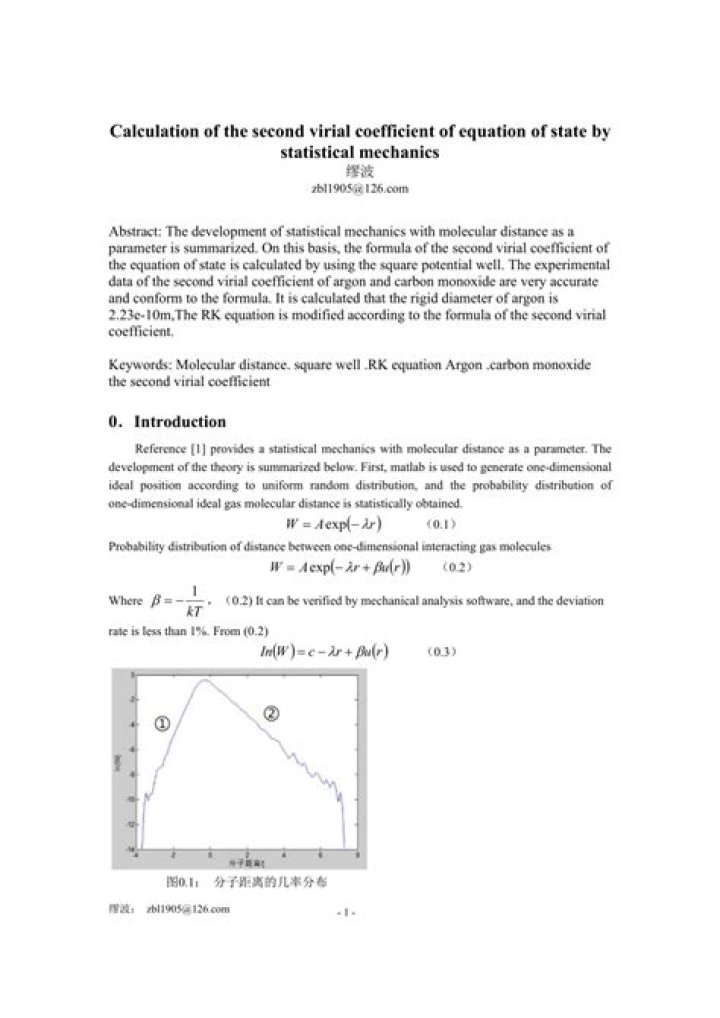
How do you calculate second virial coefficient?
(P + an2/V2)(V – nb) = nRT ; This equation relates the second-virial coefficient (B) of a gas to the van der Waals constants (a,b): B = b – a/RT , and predicts that the second virial coefficient of a gas should be negative at low temperatures, becoming less negative and possibly positve with increasing temperature.
What is the second virial coefficient?
The second virial coefficient describes the contribution of the pair-wise potential to the pressure of the gas. The third virial coefficient depends on interactions between three molecules, and so on and so forth.
What is the Berthelot equation?
It is given by:PV=RT[1+9PT c(1−6T c 2/T 2)/128P c T], where P is the pressure, V is the volume, R is the gas constant, T is the thermodynamic temperature, and T c and P c are the critical temperature and pressure of the gas.
What is the value of 2nd virial coefficient at Boyle temperature?
zero
The temperature at which second virial coefficient of a real gas is zero is called. At Boyle temperature, the gas shows ideal behaviour, i.e., PV=RT which is so only when A=RT and B=0.
What is virial equation state?
The virial Equation of state is a model that attempts to describe the properties of a real gas. This temperature is known as the Boyle temperature, TB, and it is the temperature at which the repulsive forces between the gas molecules exactly balance the attractive forces between the gas molecules.
What is a virial equation of state?
How do you use the Beattie Bridgeman equation?
It is given by P=RT(1 – ϵ)(V+B)/V 2 – A/V 2, where P is the pressure, T is the thermodynamic temperature, V is the volume, R is the gas constant, and A, B, and ϵ are constants related to five empirical constants A 0, B 0, a, b, and c by: A=A 0(1 – a/V), B=B 0(1 – b/V), and ϵ=c/VT 3.
What is the formula of Boyle temperature?
The value of Boyle’s temperature for a real gas is (TB=Rba).
What is Boyle temperature formula?
What is meaning of virial coefficient?
Definition of virial coefficient : one of the coefficients in a series of terms involving inverse powers of specific volume whose sum represents the product of specific volume by pressure for a pure gas useful form of the equation of state of a real gas is pv = A + B/v + C/v2 + …, where A, B, C, etc.



