How do you find ionic charge?
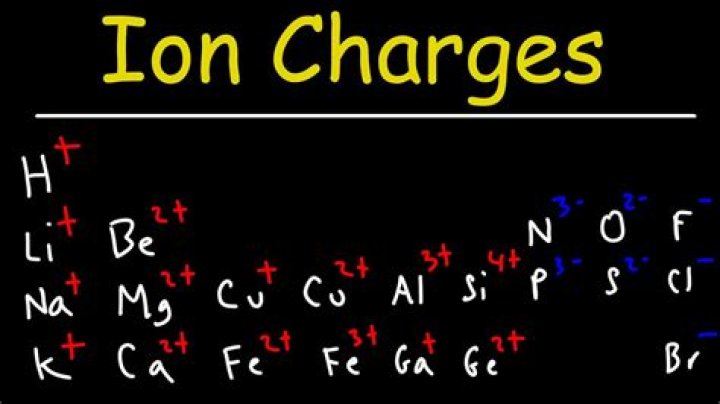
To find the ionic charge of an element you’ll need to consult your Periodic Table. On the Periodic Table metals (found on the left of the table) will be positive. Non-metals (found on the right) will be negative.
What element has a charge of negative 3?
Table of Common Element Charges
| Number | Element | Charge |
|---|---|---|
| 2 | helium | 0 |
| 3 | lithium | 1+ |
| 4 | beryllium | 2+ |
| 5 | boron | 3-, 3+ |
What is the charge of Group 2?
+2 charge
The alkali earth metals (group 2), such as, Mg or Sr lose two e-‘s to attain the configuration of Ne. Thus, the group 2 metals tend to have a +2 charge.
How do you find formal charge?
Since a chemical bond has two electrons, the “number of bonding electrons divided by 2” is by definition equal to the number of bonds surrounding the atom. So we can instead use this shortcut formula: Formal Charge = [# of valence electrons on atom] – [non-bonded electrons + number of bonds].
Is sulfur positive or negative?
Sulfur is in group 6 of the periodic table. What is the charge on its ions, and is the charge positive or negative? The charge is negative, since sulfur is a non-metal. The charge on the ion is (8 – 6) = 2.
What is the sum of negative and positive charges called?
ions
If an atom or group gains electrons or loses protons, it will have a net negative charge and is called an anion. Since ordinary matter is electrically neutral, ions normally exist as groups of cations and anions such that the sum total of positive and negative charges is zero.
Which group has elements with a 3 charge?
The main-group metals usually form charges that are the same as their group number: that is, the Group 1A metals such as sodium and potassium form +1 charges, the Group 2A metals such as magnesium and calcium form 2+ charges, and the Group 3A metals such as aluminum form 3+ charges.
What is the charge of SN?
Group IV A (14) metals form cations with +4 charge, although tin (Sn) and lead (Pb) can form cations having +2 charge.
What is the best formal charge?
0
It is best to have a formal charge of 0 for as many of the atoms in a structure as possible.
What is the formal charge of H?
zero
A hydrogen atom is made up of one proton and one electron. The formal charge of the atom, the sum of the charge of the proton and the charge of the electron, is zero. The formal charge on any atom is zero when the number of protons (the atomic number) and the number of electrons that “belong” to that atom are equal.
Why does zinc have a charge of 2?
The closed shell is very stable, so changes in electrons usually happens at the 4s2 shell. This small amount of electrons means it is more likely to lose the 2 electrons rather than taking on any in a reaction. Losing all the electrons on the fourth shell would mean Zn lost two negative charges, making it the ion Zn+2.
Can zinc have a charge of 3?
Zn always forms a 2+ ion. In fact, all of the elements in that group (Group 12) forms 2+ ions. This is because after losing the 4s electrons Zn still has a a closed n=3 shell, which is a very stable electron configuration common oxidation state is +2.
Why does sulfur have a 2 charge?
Since electrons carry a negative charge, gaining electrons will result in the formation of a negatively charged ion, or anion. For each electron gained, the ion’s overall charge decreases by 1 unit, which further confirms the fact that the neutral sulfur atom gained 2 electrons to get the (2−) charge.
Is a cation positive?
A cation is a positively charged ion with fewer electrons than protons while an anion is negatively charged with more electrons than protons. Because of their opposite electric charges, cations and anions attract each other and readily form ionic compounds.
What is the charge of H?
+1
The hydrogen ion has a positive charge of 1 (i.e.) +1. We know that hydrogen has one proton and one electron. If it loses one electron to form an ion, the charge of the hydrogen ion becomes +1.
What is the charge of Group 4 elements?
Defining group 4 as the second group in the transition metals: Most transition metals usually have a 2+ ion (with some exceptions not in this group). They may also have different charges, since their missing electrons are in the d-orbital, the atoms have more options as to what will happen.



