How do you find the density of water with temperature and pressure?
Density is usually denoted using the symbol ρ , so the formula for density is: ρ = m / V . It turns out that density isn’t constant for most substances, but in fact changes when external parameters change, such as the temperature or pressure.
How does density change with temperature and pressure?
Density and pressure/temperature Density is directly proportional to pressure and indirectly proportional to temperature. As pressure increases, with temperature constant, density increases. Conversely when temperature increases, with pressure constant, density decreases.
How does density of water change with pressure?
At the sea surface, the density is about 1027 kg/m3. Density increases when pressure increases and decreases when pressure decreases. As pressure increases, the molecules of a substance come closer resulting in a higher density.
How does density of water change with temperature?
Temperature Affects Density The density of water can also be affected by temperature. When the same amount of water is heated or cooled, its density changes. When the water is heated, it expands, increasing in volume. The warmer the water, the more space it takes up, and the lower its density.
How do you find the density of hot water?
You can find the density of any substance by dividing its mass by its volume. The formula for density is: ρ=m/v, with density represented by the symbol ρ (pronounced “rho”).
What is the formula for density of water?
Just like a solid, the density of a liquid equals the mass of the liquid divided by its volume; D = m/v. The density of water is 1 gram per cubic centimeter.
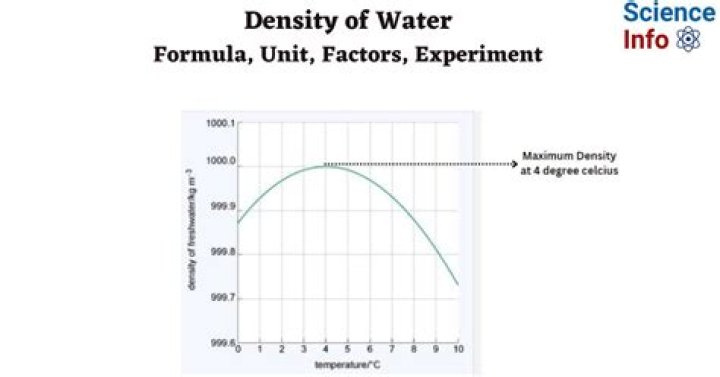
Why is water densest at 4 degrees?
As the temperature of warm water decreases, the water molecules slow down and the density increases. At 4 °C, the clusters start forming. The molecules are still slowing down and coming closer together, but the formation of clusters makes the molecules be further apart. Thus, the density of water is a maximum at 4 °C.
Why does the density of water increase as temperature decreases?
Density increase as the temperature decreases. This is the reason why liquid water is more dense than solid water. The bonds in water break more slowly as temperature decreases and the structure tend to trap fewer extra water molecules. At low temperature, more of the water has the same lattice as ice.
Does water pressure change with temperature?
As the fluid temperature increases, it tries to expand, but expansion is prevented by the walls of the container. A useful thumb rule for water is that pressure in a water-solid system will increase about 100 psi for every 1 F increase in temperature.
Does higher pressure mean higher density?
Pressure has the opposite effect on air density. Increasing the pressure increases the density.
Why does water density decrease when temperature increases?
Heating a substance causes molecules to speed up and spread slightly further apart, occupying a larger volume that results in a decrease in density. Hot water is less dense and will float on room-temperature water. Cold water is more dense and will sink in room-temperature water.
At what temperature does water density increase?
The molecules of the water are closer together, and this increases the density of the liquid. As the temperature of warm water decreases, the water molecules slow down and the density increases. At 4 °C, the clusters start forming.
How do you calculate the density of water?
Calculate the density by dividing the mass by the volume. Using the equation density = mass/volume, you can determine the density of water. Plug in the values of mass and volume you determined and solve.
How to calculate densities at various temperatures?
How to Calculate Densities at Various Temperatures Subtract Final Temperature. Subtract the final temperature in degrees Celsius from the initial temperature in degrees… Multiply Temperature Difference. Multiply this temperature difference by the volumetric temperature expansion… Find Final
What is the specific temperature of water?
Specific gravity (SG) for water is given for four different reference temperatures (39.2, 59, 60 and 68°F). From 32 to 212°F the pressure is 14.7 psi, and for temperatures >212°F, the pressure is equal to water saturation pressure.
What is the equation for the density of water?
The formula for density is d = M/V, where d is density, M is mass, and V is volume. Density is commonly expressed in units of grams per cubic centimetre. For example, the density of water is 1 gram per cubic centimetre, and Earth’s density is 5.51 grams per cubic centimetre.



