How do you find the volume of an HCP unit cell?
Hint: The volume of the unit cell can be determined by taking the product of the area and height of the cell. For the hexagonal close packing structure$(\text{HCP)}$, the volume is equal to: $\text{Area of base }\!
What is the unit cell of HCP?
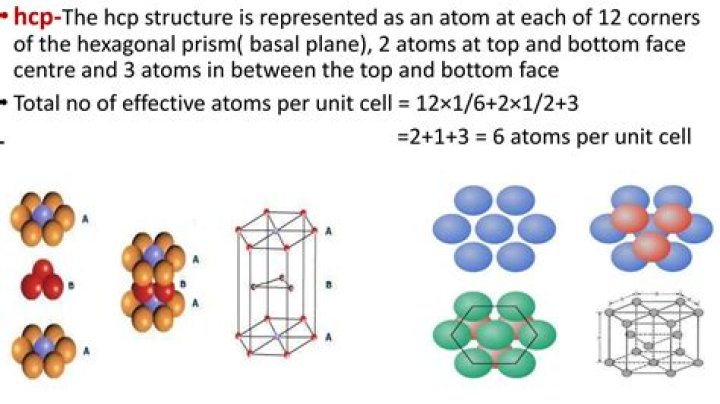
The hexagonal closest packed (hcp) has a coordination number of 12 and contains 6 atoms per unit cell. The face-centered cubic (fcc) has a coordination number of 12 and contains 4 atoms per unit cell. The body-centered cubic (bcc) has a coordination number of 8 and contains 2 atoms per unit cell.
How do you calculate C in HCP?
c a = 8 3 = 1.633 2 Page 3 2. Show that the atomic packing factor for HCP is 0.74. Now, the unit cell volume is the product of the base area times the cell height, c.
What is the volume of the unit cell in terms of a?
What is the volume of a unit cell? The unit cell volume (V) is equal to the cubed cell-edge length (a). In a face-centered cubic structure, there would be four atoms per unit cell and the nickel density in this structure would be four times as high.
How do u find the volume of a hexagon?
The formula for the volume of a hexagonal prism is, volume = [(3√3)/2]a2h cubic units where a is the base length and h is the height of the prism. We can also use the other formula V = 3abh, where a = apothem length, b = length of a side of the base, and h = height of the prism.
What is the height of an HCP unit cell?
In the calculation of packing efficiency of hcp arrangement,the height of the unit cell is taken as 4r. 2/3.
What is the volume of HCP?
162 r3.
How is HCP calculated?
- Each Corner atom of hexagonal face is shared by 6 unit cells i.e. they contribute 1/6th of the mass.
- Thus, contribution of corner atoms = 2 x 6 x 1/6 = 2.
- The atom at the center of the hexagonal faces is shared by two cells each.
- Therefore, contribution of face centered atoms = 2 x 1/2 = 1.
How is hcp calculated?
WHAT IS A in hcp?
Each atom has twelve nearest neighbors in hcp. In the ideal structure, the distance between the planes is 1.633a, where a is the distance between the atoms. For some materials that are commonly considered hcp, the distance bewteen the planes deviates from the ideal structure. Beryllium, Magnesium and Titanium form hcp.
How many atoms are in HCP?
Cell of an HCP lattice is visualized as a top and bottom plane of 7 atoms, forming a regular hexagon around a central atom.
How many atoms in HCP unit cell?
The hexagonal closest packed (hcp) has a coordination number of 12 and contains 6 atoms per unit cell. The face-centered cubic (fcc) has a coordination number of 12and contains 4 atoms per unit cell. The body-centered cubic ( bcc ) has a coordination number of 8 and contains 2atoms per unit cell.
What is hexagonal unit cell?
Hexagon unit cell is the unit cell of a hexagonal crystal system. It represents the arrangement of atoms in a material with a hexagon crystal structure. It has two axes with a similar length and one axis with a different length.
What is a HCP number?
Applicant Area. The HCP Number is a unique number assigned to each HCP physical location by RHCD. The HCP Number serves as RHCD’s tool to track each HCP’s application throughout the process. The HCP number must be used when completing all forms and should be written on all paper documentation submitted to RHCD.
What is an unit cell?
unit cell. n. The smallest building block of a crystal, consisting of atoms, ions, or molecules, whose geometric arrangement defines a crystal’s characteristic symmetry and whose repetition in space produces a crystal lattice. unit cell. u′nit cell`. n. the simplest unit of a regular crystal lattice.



