How do you know if a cyclohexane is cis or trans?
Cis-trans Isomerism in Cycloalkanes A cycloalkane has two distinct faces, and any substituent on a ring lies toward one of two faces. When two substituents on a ring point to the same face, they are cis. When the two substituents point to opposite faces, they are trans.
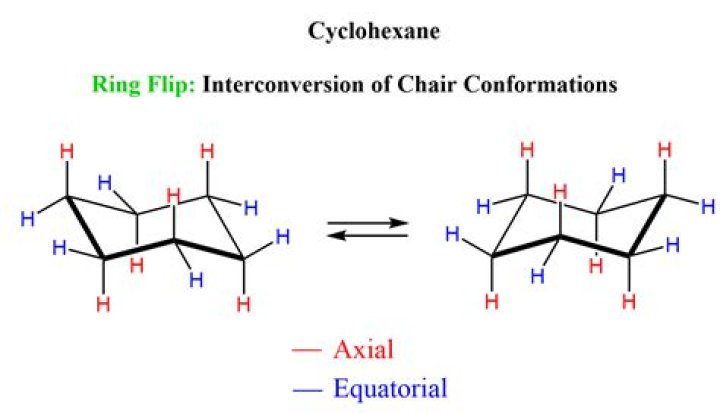
Which is a chair conformation of cyclohexane?
The most stable conformation of cyclohexane is called “chair“ conformation, since it somewhat resembles a chair. In the chair conformation of cyclohexane, all the carbons are at 109.5º bond angles, so no angle strain applies.
Is cis or trans chair conformation more stable?
The diaxial trans conformer has 3.6 kcal of steric strain, of course and is much less favored than the diequatorial conformer. The cis isomer is less stable than the trans because in it, one methyl must be axial. The cis isomer is therefore less stable than the trans by 1.8 kcal.
Is chair form of cyclohexane planar?
Cyclohexane can obtain a partially plane conformation called “half chair” but with only with excessive amounts of ring strain. The half chair conformation is formed by taking planar cyclohexane and lifting one carbon out of the plane of the ring.
What is trans cyclohexane?
What is Trans Cyclohexane? Trans cyclohexane is a geometric isomer of cyclohexane which has its substituents in the opposite planes. That means; if one substituent is in the equatorial plane, the other substituent will be in the axial plane and vice versa.
Is trans more stable than cis cyclohexane?
You should find that the trans isomer of 1,4-dimethylcyclohexane is more stable than the cis isomer. The relative stabilities of the cis and trans isomers of disubstituted cyclohexanes depends upon which isomer has the most stable conformer. The most stable isomer for disubstituted cyclohexanes is summarized below.
Which cyclohexane is most stable?
chair conformation
The chair conformation is the most stable conformer. At 25 °C, 99.99% of all molecules in a cyclohexane solution adopt this conformation.
What makes a cyclohexane CIS?
Cis cyclohexane is a geometric isomer of the organic compound cyclohexane. The cyclohexane molecule should have two (or more) substituents in order to show this isomerism. If the two substituents groups are in the same plane (either equatorial or axial), then we call it the cis isomer of cyclohexane.



