How do you name ionic compounds and acids?
In simple binary acids, one ion is attached to hydrogen. Names for such acids consist of the prefix “hydro-“, the first syllable of the anion, and the suffix “-ic”. Complex acid compounds have oxygen in them. For an acid with a polyatomic ion, the suffix “-ate” from the ion is replaced with “-ic.”
Can ionic compounds be acids?
Acidity/basicity Ionic compounds containing hydrogen ions (H+) are classified as acids, and those containing electropositive cations and basic anions ions hydroxide (OH−) or oxide (O2−) are classified as bases. Other ionic compounds are known as salts and can be formed by acid–base reactions.
What are the three rules for naming acids?
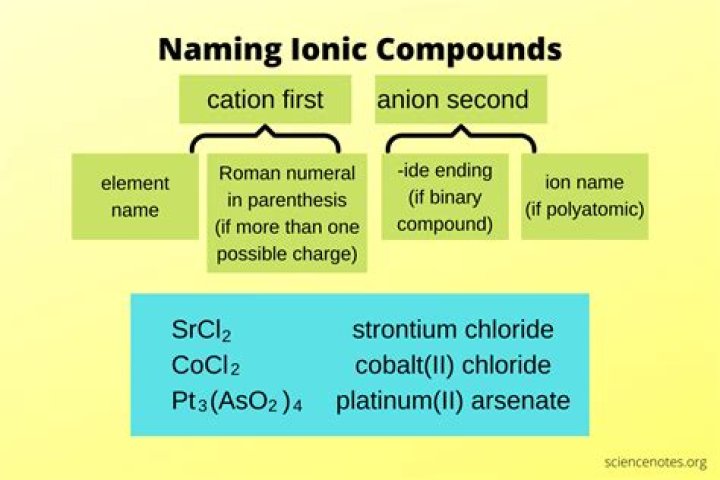
Example
- When the anion ends in –ide, the acid name begins with the prefix hydro-.
- When the anion ends in –ate, the name of the acid is the root of the anion followed by the suffix –ic.
- When the anion ends in –ite, the name of the acid is the root of the anion followed by the suffix –ous.
What are the rules in naming ionic and molecular compounds?
A diatomic compound (or diatomic molecule) contains two atoms, which may or may not be the same. Metals combine with nonmetals to give ionic compounds. When naming binary ionic compounds, name the cation first (specifying the charge, if necessary), then the nonmetal anion (element stem + -ide).
When naming an ionic compound What comes first?
For binary ionic compounds (ionic compounds that contain only two types of elements), the compounds are named by writing the name of the cation first followed by the name of the anion. For example, KCl, an ionic compound that contains K+ and Cl- ions, is named potassium chloride.
What are the importance of naming compounds?
The primary function of chemical nomenclature is to ensure that a spoken or written chemical name leaves no ambiguity concerning which chemical compound the name refers to: each chemical name should refer to a single substance.
How do you name and write ionic compounds?
How Ionic Compounds Are Named. You first write the name of the metal, lithium, and then write the name of the nonmetal, adding an -ide ending so that sulfur becomes sulfide. Ionic compounds involving polyatomic ions follow the same basic rule: Write the name of the metal first, and then simply add the name of the nonmetal (with the polyatomic anions,…
What are some simple rules for naming ionic compounds?
The following rules apply for naming ionic compounds. A simple cation has the same name as its parent element. The cation is always named first and the anion second. A simple cation takes its name from the name of the element. A simple anion is named by taking the first part of the element name and adding -ide.
Which ones are ionic compounds?
In ionic compounds the constituent elements exist as ions. Ions are atoms or groups of atoms that carry a charge by virtue of losing or gaining one or more electrons. Ionic compounds result from the combination of a positive ion known as a cation and a negative ion called an anion.
What are These ionic compound names?
Fe 2+Ferrous



