How do you synthesis epoxides?
Epoxides (also known as oxiranes) are three-membered ring structures in which one of the vertices is an oxygen and the other two are carbons. The most important and simplest epoxide is ethylene oxide which is prepared on an industrial scale by catalytic oxidation of ethylene by air.
Which reagent is used to prepare an epoxide?
Epoxidation is the chemical reaction which converts the carbon–carbon double bond into oxiranes (epoxides), using a variety of reagents including air oxidation, hypochlorous acid, hydrogen peroxide, and organic peracid (Fettes, 1964).
What reaction forms epoxide?
This got some friendly flak from an inorganic chemist in the crowd, but for our purposes, it’s true. We only cover one reaction: how to cleave ethers with strong acid. To review, here’s what this reaction looks like for several different ethers.
Which of the following reagent can convert epoxide ring into alcohol?
Epoxidized linseed oil (Edenol B316)
| PARAMETER | UNIT |
|---|---|
| HMIS personal protection | chemical-resistant gloves, eye protection |
| USE & PERFORMANCE | |
| Manufacturer | Emery Oleochemicals |
| Recommended for polymers | PVC & its copolymers, chlorinated rubber, CN |
How do you break an epoxide ring?
Epoxides can also be opened by other anhydrous acids (HX) to form a trans halohydrin. When both the epoxide carbons are either primary or secondary the halogen anion will attack the less substituted carbon and an SN2 like reaction.
How do you break open epoxide?
Epoxides can also be opened by other anhydrous acids (HX) to form a trans halohydrin. When both the epoxide carbons are either primary or secondary the halogen anion will attack the less substituted carbon and an S N2 like reaction.
Does epoxide react with alcohol?
Protonation makes the epoxide more electrophilic and creates a better leaving group. The leaving group is the protonated oxygen atom of the epoxide in the form of a neutral alcohol. Typically the nucleophile is then deprotonated to give a neutral product.
How do epoxide rings open?
Both strong and weak nucleophiles open the epoxide ring by an opposite-side nucleophilic attack. This puts the nucleophile and the alkoxy group of opposite sides and as a result, trans or anti-products are always formed.
What breaks an epoxide?
Epoxides are much more reactive than simple ethers due to ring strain. Nucleophiles attack the electrophilic C of the C-O bond causing it to break, resulting in ring opening. Opening the ring relieves the ring strain.
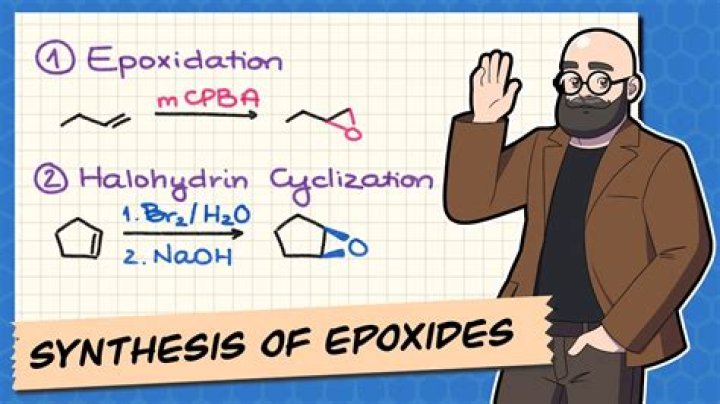
What is synthesis of epoxides?
Synthesis of epoxides. A chiral bisaryl-silyl-protected pyrrolidine acts as a very selective epoxidation organocatalyst using simple oxidation agents. The scope of the reaction is demonstrated by the formation of optically active α,β-epoxy aldehydes in high yields and enantioselectivities.
What are peroxide-forming chemicals?
Peroxide-forming chemicals (PFs) are chemicals that can “auto-oxidize” with atmospheric oxygen under ambient conditions to form organic peroxides (contains an -O–O- bond). Peroxide formation can be initiated by exposure to air, self-polymerization, or solvent impurities.
What is hydrogen peroxide epoxidation of olefins?
An efficient epoxidation of a broad range of olefins using hydrogen peroxide as the oxidant has been accomplished in the presence of acetic acid and a manganese catalyst that exhibits an uncommon chemoselectivity. I. Garcia-Bosch, X. Ribas, M. Costas, Adv. Synth.
What is an intramolecular epoxide formation reaction?
An intramolecular epoxide formation reaction is one of the key steps in the Darzens reaction . In the Johnson–Corey–Chaykovsky reaction epoxides are generated from carbonyl groups and sulfonium ylides. In this reaction, a sulfonium is the leaving group instead of chloride.



