How does free energy relate to equilibrium?
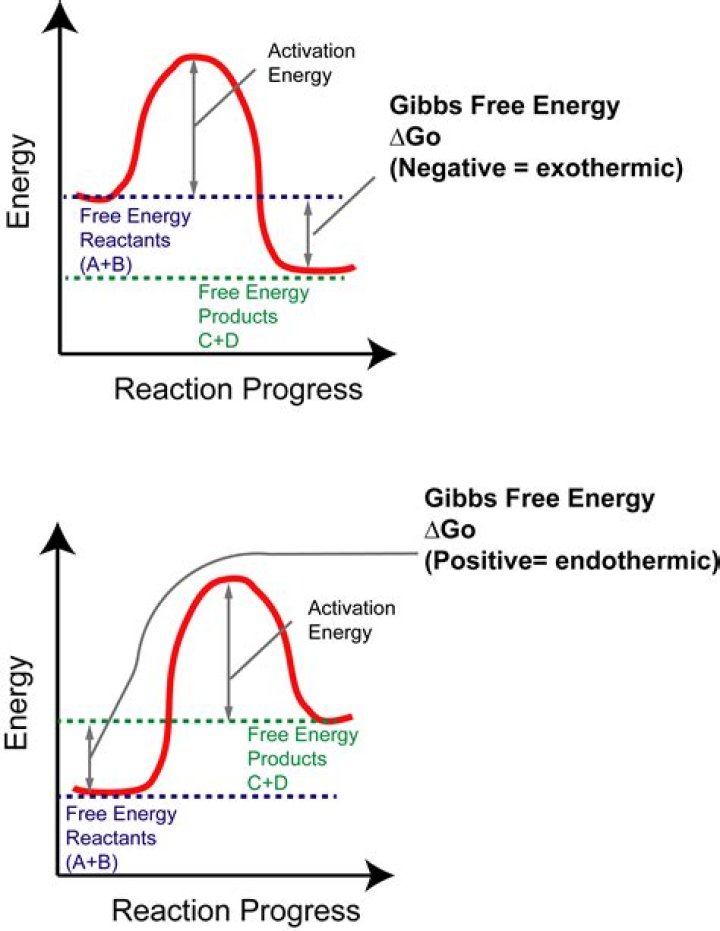
The balance between reactants and products in a reaction will be determined by the free energy difference between the two sides of the reaction. The greater the free energy difference, the more the reaction will favor one side or the other.
What is the Gibbs free energy at equilibrium?
The Gibbs energy G is a quantity that becomes more negative during the course of any natural process. Thus as a chemical reaction takes place, G only falls and will never become more positive. At this point G is at a minimum (see below), and no further net change can take place; the reaction is then at equilibrium.
What is the relationship between equilibrium constant and Gibbs free energy?
The relation between Gibbs energy and equilibrium constant is △Go=−RTlnKeq. The relation between Gibbs energy and change in enthalpy is △G= △H- T△S. Note: The beauty of △G= △H- T△S is the ability to determine the relative importance of the enthalpy and entropy as driving forces behind a reaction.
Why is Gibbs free energy 0 at equilibrium?
Re: Delta G=0 at equilibrium Gibbs free energy is the energy free to do work. At equilibrium, no net energy is being gained or lost (nothing is really changing). Thus, at equilibrium, Delta G is 0.
How do you find the equilibrium constant from thermodynamic data?
The relation K=exp(−ΔrG∘/RT) (Eq. 11.8. 11) gives us a way to evaluate the thermodynamic equilibrium constant K of a reaction at a given temperature from the value of the standard molar reaction Gibbs energy ΔrG∘ at that temperature. If we know the value of ΔrG∘, we can calculate the value of K.
When Gibbs free energy is minimum?
So, the Gibbs free energy change will reach a minimum value when the change of entropy equals to zero. Actually, for chemical process calculations, the chemist are interseted on the thermodynamic changes. So, the Gibbs free energy change will reach a minimum value when the change of entropy equals to zero.
What is the meaning of the standard free energy change ∆ G as compared with ∆ G?
∆G is the change of Gibbs (free) energy for a system and ∆G° is the Gibbs energy change for a system under standard conditions (1 atm, 298K). Where ∆G is the difference in the energy between reactants and products. In addition ∆G is unaffected by external factors that change the kinetics of the reaction.
Is Delta G naught zero at equilibrium?
In short, it is ∆G (NOT ∆G°) that will be zero at equilibrium and the sign of it (generated by the combination of ∆G° and RT ln Q in Equation #2), will define which way the reaction proceeds.
What is the difference between thermodynamic equilibrium and non-equilibrium?
Thermodynamic equilibrium. In non-equilibrium systems, by contrast, there are net flows of matter or energy. If such changes can be triggered to occur in a system in which they are not already occurring, it is said to be in a metastable equilibrium.
Is thermodynamic free energy absolute or relative?
Thermodynamic free energy. Since free energy usually contains potential energy, it is not absolute but depends on the choice of a zero point. Therefore, only relative free energy values, or changes in free energy, are physically meaningful.
What is maximum and minimum at thermodynamic equilibrium?
For a completely isolated system, S is maximum at thermodynamic equilibrium. For a system with controlled constant temperature and volume, A is minimum at thermodynamic equilibrium. For a system with controlled constant temperature and pressure, G is minimum at thermodynamic equilibrium. The various types of equilibriums are achieved as follows:
What happens to the free energy during a chemical reaction?
The total free energy of the system (reactants + products) always becomes more negative as the reaction proceeds. Eventually it reaches a minimum value at a system composition that defines the equilibrium composition of the system, after which time no further net change will occur.



