How does vapor pressure affect heat of vaporization?
At a pressure greater than 1 atm, water boils at a temperature greater than 100°C because the increased pressure forces vapor molecules above the surface to condense. Hence the molecules must have greater kinetic energy to escape from the surface. Conversely, at pressures less than 1 atm, water boils below 100°C.
Does vapor pressure increase with enthalpy of vaporization?
The vaporization curves of most liquids have similar shapes with the vapor pressure steadily increasing as the temperature increases (Figure 1). where ΔHvap is the Enthalpy (heat) of Vaporization and R is the gas constant (8.3145 J mol-1 K-1).
What is the relationship between vapor pressure and evaporation rate?
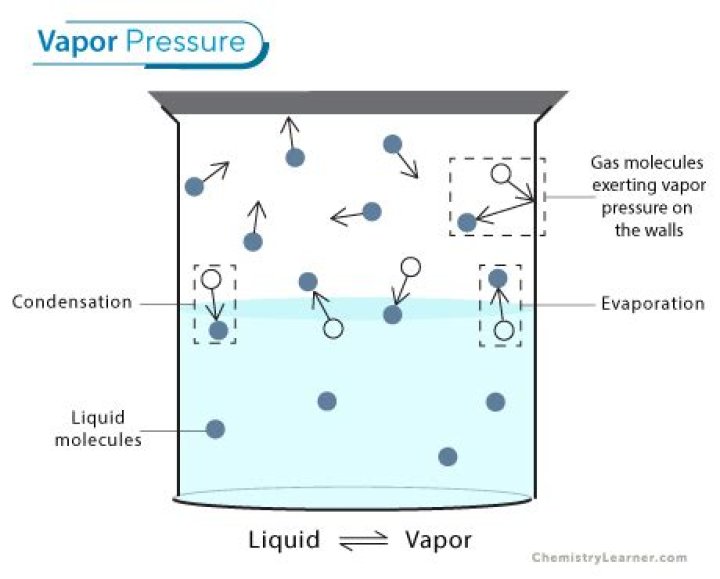
Summary. Vapor pressure is a measure of the pressure exerted by a gas above a liquid in a sealed container. Strong intermolecular forces produce a lower rate of evaporation and a lower vapor pressure. Weak intermolecular forces produce a higher rate of evaporation and a higher vapor pressure.
How do you find heat vaporization from vapor pressure?
If the problem provides the two pressure and two temperature values, use the equation ln(P1/P2)=(Hvap/R)(T1-T2/T1xT2), where P1 and P2 are the pressure values; Hvap is the molar heat of vaporization; R is the gas constant; and T1 and T2 are the temperature values.
How can you increase the rate of vaporization?
The presence of a breeze, a powerful wind, or some other form of air circulation can speed up this process and make the environment of the liquid less humid. Therefore, by decreasing the humidity of the liquid’s surrounding, a powerful breeze or wind can increase the rate at which the liquid evaporates.
What causes vapor pressure to increase?
As the temperature of a liquid increases, the kinetic energy of its molecules also increases. As the kinetic energy of the molecules increases, the number of molecules transitioning into a vapor also increases, thereby increasing the vapor pressure.
Under which conditions will vaporization best occur?
Heat must be supplied to a solid or liquid to effect vaporization. If the surroundings do not supply enough heat, it may come from the system itself as a reduction in temperature.
How does heat of vaporization change with temperature?
The heat of vaporization diminishes with increasing temperature and it vanishes completely at a certain point called the critical temperature (Critical temperature for water: 373.946 °C or 705.103 °F, Critical pressure: 220.6 bar = 22.06 MPa = 3200 psi ).
Why does vapor pressure increase with temperature?
Does vapor pressure change with pressure?
And in all this, regardless of what atmospheric pressure we are at, the shape of the liquid-vapor coexistence curve does not change. The vapor pressure is hardly influenced by the atmospheric pressure in that regard, because we ignore the intermolecular forces in ideal vapors.
What is the HVAP constant of heat vaporization?
Hvap is the change in heat vaporization, R is the universal gas constant, which is (8. 31 J/mol•K), T is the absolute, or Kelvin, temperature, and C is the constant that is not related to heat capacity. Haven’t found the relevant content? Hire a subject expert to help you with Vapor Pressure and Heat Evaporation Lab Report
What is the relationship between vapor pressure and temperature?
In mathematical terms, the relationship between the vapor pressure of a liquid and temperature is described in the Clausius-Clayperon equation,
What happens to gas molecules during evaporation?
During the evaporation process of the liquid, new gas molecules exert pressure in the sealed container, while some of the gas condenses back to the liquid state. If the temperature inside the container is kept constant, then the equilibrium at some point will be reached.
Why do the results of an evaporation experiment vary?
Discussion At the end of this experiment, the results we obtained varied because of the different temperatures and pressures that we observed. During the evaporation process of the liquid, gas molecules exert pressure in the sealed container, while some of the gas condenses back to the liquid state.



