How many outer shell electrons does lead have?
4 electrons
Lead is the chemical element which has atomic number 82 AND symbol “Pb”. In case of lead, there are 4 electrons present in the 6th or outer shell. A number of valence electrons are defined as a number of electrons present in the outer shell of atom or molecule.
How many valence electrons does tin and lead have?
four valence electrons
Group 4A (or IVA) of the periodic table includes the nonmetal carbon (C), the metalloids silicon (Si) and germanium (Ge), the metals tin (Sn) and lead (Pb), and the yet-unnamed artificially-produced element ununquadium (Uuq). The Group 4A elements have four valence electrons in their highest-energy orbitals (ns2np2).
Is there tin in lead?
The alloys of tin and lead have characteristic weight percentages. Ideally, solder is 63 percent tin and 37 percent lead. However, it is usually sold as 60 percent tin and 40 percent lead. Terne plate tends to be 75 percent lead and 25 percent tin.
How many electrons does tin have?
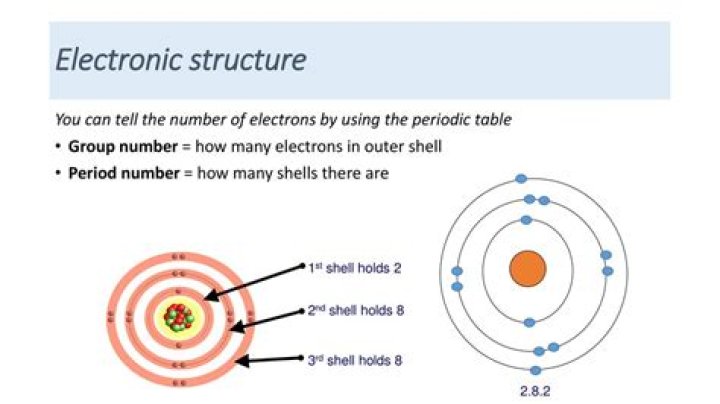
2, 8, 18, 18, 4
Tin/Electrons per shell
How many shells does the lead atom have?
Data Zone
| Classification: | Lead is an ‘other metal’ |
|---|---|
| Electrons: | 82 |
| Protons: | 82 |
| Neutrons in most abundant isotope: | 126 |
| Electron shells: | 2,8,18,32,18,4 |
What is the outer electron configuration for lead?
Lead Atomic and Orbital Properties
| Atomic Number | 82 |
|---|---|
| Number of Neutrons | 125 |
| Shell structure (Electrons per energy level) | [2, 8, 18, 32, 18, 4] |
| Electron Configuration | [Xe] 4f14 5d10 6s2 6p2 |
| Valence Electrons | 6s2 6p2 |
What is the valence of lead?
4 valence electrons
Lead has 4 valence electrons.
What’s the difference between tin and lead?
Lead and tin are metallic elements. The chemical symbol for lead is Pb, and the chemical symbol for tin is Sn. The key difference between lead and tin is that lead appears in metallic grey colour with a blue hue whereas tin appears as a silvery-white metal with a faint yellow hue.
How many electrons does lead have?
2,8,18,32,18,4
Lead/Electrons per shell
How many neutrons does tin have?
69 neutrons
Example: This tin atom has 50 protons, 69 neutrons and 48 electrons.
Is tin toxic to humans?
Tin has no known natural biological role in living organisms. It is not easily absorbed by animals and humans. The low toxicity is relevant to the widespread use of tin in dinnerware and canned food. Nausea, vomiting and diarrhea have been reported after ingesting canned food containing 200 mg/kg of tin.
What is the number of electrons in a tin atom?
Tin has an atomic number of #50#, so right from the start you know that its electron configuration must account for a total of #50# electrons. The fifth energy level, #n = color(blue)(5)#, is tin’s outermost energy shell.
What is the oxidation number of lead in lead?
lead does have an oxidation state of 4+ and elements above such as carbon and tin also have 2+ as an oxidation state. Just look at the electron configuration : the outer [math]s^2 p^2 [/math] orbitals will be able to lose 2 or even 4 electrons, or even gain 4 electrons.
Is tin a metal or nonmetal?
Tin is a post-transition metal in group 14 of the periodic table. It is obtained chiefly from the mineral cassiterite, which contains tin dioxide. The first alloy used on a large scale was bronze, made of tin and copper, from as early as 3000 BC. Tin is a chemical element with atomic number 50 which means there are 50 protons in its nucleus.
Where does tin come from on the periodic table?
Tin is a post-transition metal in group 14 of the periodic table. It is obtained chiefly from the mineral cassiterite, which contains tin dioxide. The first alloy used on a large scale was bronze, made of tin and copper, from as early as 3000 BC.



