How many particles are in a gram?
1.4. The definition of Avogadro’s number of 6.022 × 1023/mole is the number of atoms or molecules per one gram atomic weight.
How do you calculate number of particles?
The mathematical equation, N = n × NA, can be used to find the number of atoms, ions or molecules in any amount (in moles) of atoms, ions or molecules: 10 moles of helium atoms = 10 × (6.022 × 1023) = 6.022 × 1024 helium atoms.
How do you convert between mass and number of particles?
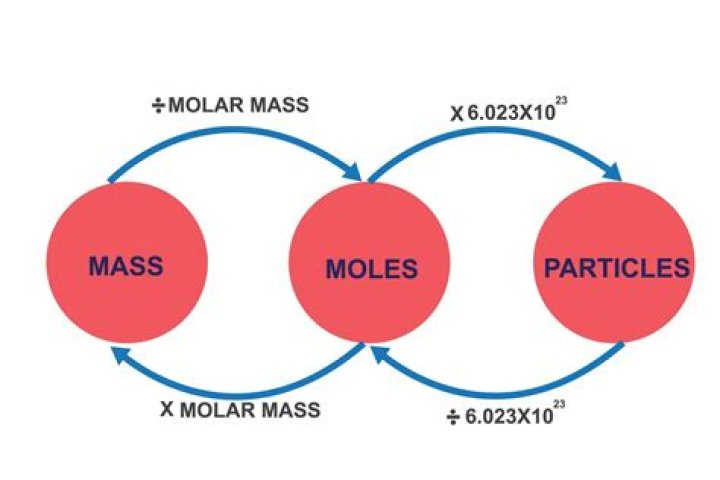
In order to convert between mass and amount (of atoms or molecules), we use a mole. From the periodic table, we know the mass of each element in one mole of that element. And, we know how many atoms are in one mole. This is equal to Avogadro’s number (6.02 x 10^23).
How do you calculate grams to particles?
To calculate the number of atoms in a sample, divide its weight in grams by the amu atomic mass from the periodic table, then multiply the result by Avogadro’s number: 6.02 x 10^23.
How many grams are contained in 1 gram atom of any?
Answer Expert Verified 1 gram atom means 1 mole of that element, i.e., the atomic mass expressed in grams. Hence, it can be easily be deduced that 1 gram atom of sodium will be 23 grams.
Is the number of particles per unit weight?
The expression particle density refers to the number of particles per unit volume, not to the density of a single particle, and it is usually expressed as n.
What is a gram formula weight and how is it used?
The atomic mass of one mole of an element, molecular compound or ionic compound is known as the Gram formula mass (a.k.a. molar mass). Thus mole is a fundamental quantity when chemical calculations are involved. This concept is used extensively across organic, inorganic, and physical chemistry.
How many grams are contained in 2 gram atoms of Na?
23 g
23 g.
How do you convert from molecules to grams?
To convert from molecules to grams, it is necessary to first convert the number of molecules of a substance by dividing by Avogadro’s number to find the number of moles, and then multiply the number of moles by the molar mass of this substance.
How do you calculate grams?
Multiply the number of grams by the conversion number. Look on the label of the food your want to calculate the calories for. Each nutrient will be listed in grams. Once you find the one you are looking for, you can multiply that number by the number of calories that specific nutrient has in each gram.
How to convert grams to molecules?
(See explanation) To convert grams to moles, you will need to molar mass of that specific element, molar mass is found my multiplying the number of atoms present in the compound by the relative atomic mass found on the bottom of the element block.
What is the formula for converting atoms to grams?
Converting gram to atom or converting atom to gram is an basic and essential process to solve difficult calculations in chemistry. The conversion requires Avogadro ‘s number, mole of substance and atomic weight. Formula for conversion of gram to atoms are given below. Formula to convert grams to moles : moles = $\\frac{atoms}{Avogadro’s \\ number}$.



