How many protons neutrons and electrons does silver 108 have?
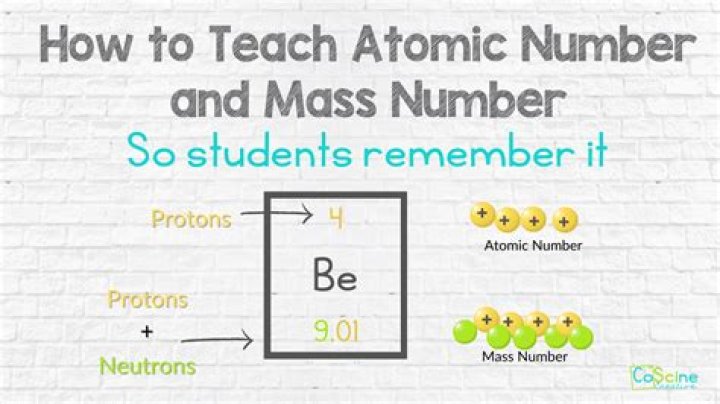
mass number is 108, neutrons = 108 – 47 protons = 61 neutrons. Ag+ = 47 electrons – 1 electron = 46 electrons. Finally: 47 protons , 61 neutrons and 46 electrons.
How many neutrons are in the nucleus of silver 108?
Heat of vaporization: 250.580 kJ/mol. Number of Protons/Electrons: 47. Number of neutrons: 61.
What is an atomic number Answer Key?
atomic number, the number of a chemical element in the periodic system, whereby the elements are arranged in order of increasing number of protons in the nucleus. Accordingly, the number of protons, which is always equal to the number of electrons in the neutral atom, is also the atomic number.
How many protons and neutrons do silver have?
47
Silver/Atomic number
How many protons neutrons and electrons does silver 109 have?
Ag has 47 electrons, also = 47 protons and therefore 108 – 47 = 61 neutrons. Since number of protons and neutrons rarely changes, Ag 109 will have 47 protons and 61 neutrons.
What are the respective charges of protons neutrons and electrons?
Proton—positive; electron—negative; neutron—no charge. The charge on the proton and electron are exactly the same size but opposite. The same number of protons and electrons exactly cancel one another in a neutral atom.
How many protons neutrons and electrons are in ag 107?
Ag-107: 47 protons and 60 neutrons →10747Ag. Ag-109: 47 protons and 62 neutrons →10947Ag.
What is the mass number of silver 108?
107.90595
Silver-108
| PubChem CID | 167088 |
|---|---|
| Structure | Find Similar Structures |
| Molecular Formula | Ag |
| Synonyms | Silver-108 14391-65-2 Silver, isotope of mass 108 108Ag Silver 108 More… |
| Molecular Weight | 107.90595 |
How many protons and electrons does silver have?
Silver – Mass Number – Neutron Number – Ag 2020-11-21 by Nick Connor Silver is a chemical element with atomic number 47 which means there are 47 protons and 47 electrons in the atomic structure. The chemical symbol for Silver is Ag.
What is the electron configuration and density of silver?
In the periodic table, the elements are listed in order of increasing atomic number Z. Electron configuration of Silver is [Kr] 4d10 5s1. Possible oxidation states are +1. Density of Silver is 10.49g/cm3. Typical densities of various substances are at atmospheric pressure.
What is the atomic mass of silver?
Atomic Mass of Silver Element Silver Atomic Number 47 Symbol Ag Element Category Transition Metal Phase at STP Solid
What is the number of protons and electrons in gold?
Iridium has 77 protons, 115 neutrons and 77 electrons: 78: Platinum has 78 protons, 117 neutrons and 78 electrons: 79: Gold has 79 protons, 118 neutrons and 79 electrons: 80: Mercury has 80 protons, 121 neutrons and 80 electrons: 81: Thallium has 81 protons, 123 neutrons and 81 electrons: 82: Lead has 82 protons, 125 neutrons and 82 electrons: 83



