Is breaking bonds always endothermic?
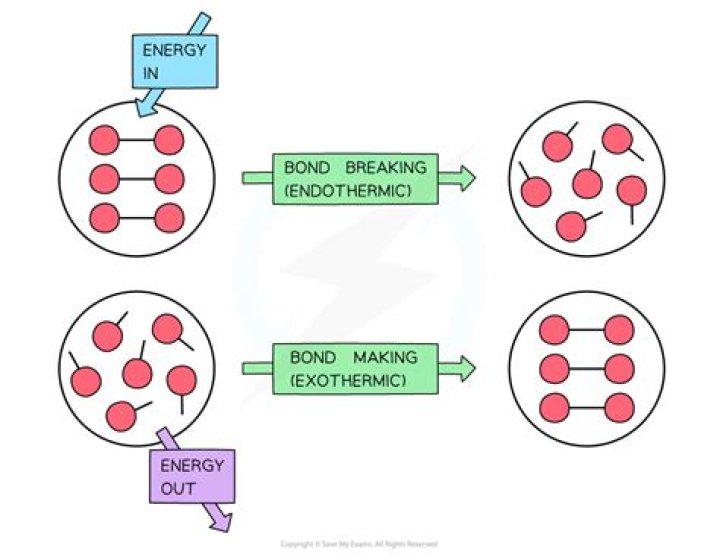
Energy is absorbed to break bonds. Bond-breaking is an endothermic process. Bond-making is an exothermic process. Whether a reaction is endothermic or exothermic depends on the difference between the energy needed to break bonds and the energy released when new bonds form.
Is breaking of bond exothermic or endothermic?
BREAKING AND FORMING BONDS Bond breaking is an endothermic process, because it requires energy. Bond forming is an exothermic process, because it releases energy.
Why are bond breaking processes always endothermic?
A reaction to break a bond always requires the input of energy and so such a process is always endothermic. When atoms come together to form new chemical bonds, the electrostatic forces bringing them together leave the bond with a large excess of energy (usually in the form of vibrations and rotations).
Why is bond breaking exothermic?
Breaking bonds between atoms requires energy. Creating new bonds releases it. For this reaction, the energy released is larger than the energy absorbed. This means combustion has an overall negative enthalpy and is an exothermic reaction.
Is bond breaking positive or negative?
Generally, a positive change in enthalpy is required to break a bond, while a negative change in enthalpy is accompanied by the formation of a bond. In other words, breaking a bond is an endothermic process, while the formation of bonds is exothermic.
Why is breaking bonds exothermic?
Is breaking bonds positive or negative?
Why do bonds break?
When a chemical reaction occurs, molecular bonds are broken and other bonds are formed to make different molecules. For example, the bonds of two water molecules are broken to form hydrogen and oxygen. Energy is always required to break a bond, which is known as bond energy. Energy is always required to break a bond.
Why does breaking bonds absorb energy?
The reason there is energy released in the process is because the products formed (ADP and hydrogenphosphate/phosphate) have stronger covalent bonds (plus intermolecular forces with the surrounding solution and dissolved ions) than the starting materials. This is the case for any exothermic process.
What happens to bonds in an endothermic reaction?
Chemical reactions that absorb (or use) energy overall are called endothermic. In endothermic reactions, more energy is absorbed when the bonds in the reactants are broken than is released when new bonds are formed in the products.
Is bond formation always exothermic?
Exothermic reaction: The type of reaction in which the reaction releases heat, the reaction is known as exothermic reaction. As the formation of bonds increases stability then the reactions are exothermic in nature. Hence, bond formation is always an exothermic reaction.
What is absorbed during bond breaking but liberated during bond formation?
In endothermic reactions, more energy is absorbed when the bonds in the reactants are broken than is released when new bonds are formed in the products.
Is bond breaking endothermic or exothermic?
$\\begingroup$ @JosephHirsch, Reactions may be endothermic or exothermic, but as a general rule, bond-breaking is indeed endothermic (requires energy applied to break the bonds) whereas bond-making is exothermic (outputs energy), as you said.
Does breaking of bonds release energy?
Bond breaking does NOT release energy!! In the course of a chemical reaction, you typically break some bonds and form some new bonds. The bond breaking is always endothermic. The formation of new bonds is exothermic, so depending on whether the old bonds or the new bonds were stronger, the reaction overall can be either endothermic…
How to determine if a reaction is exothermic or endothermic?
If the total energy required to break bonds in the reactants is less than the total energy released when new bonds are formed in the products, it is an exothermic reaction. Alternatively, you can denote: Er E r = Total energy required to break bonds in the reactants Ep E p = Total energy released when new bonds are formed in the products
What is the relationship between bond breaking and bond forming?
Bond breaking and bond forming occurs during a chemical reaction. The energy changes in chemical reactions are caused by bond breaking and bond forming. Breaking a bond is endothermic. Energy is taken in to break a chemical bond. E.g. H −H(g) → H(g)+ H(g) H − H ( g) → H ( g) + H ( g), whereby ΔH = +436 kJ mol−1 Δ H = + 436 kJ mol − 1



