Is detergent polar nonpolar or amphipathic?
Detergents are amphipathic molecules with a polar portion and a hydrophobic portion. Detergents respond to an aqueous environment following the same principles as do membrane lipids.
Is detergents polar or nonpolar?
Soap and detergents are quite unique molecules with the ability to dissolve in both polar and non-polar solvents. Both Soap and detergents are composed of long non-polar hydrocarbon molecules with a polar or ionic end, thus they combine both polar and non-polar characteristics in the same molecule.
Is detergent an amphipathic molecule?
Detergents are amphipathic molecules that contain polar or charged hydrophilic groups (heads) at the end of long lipophilic hydrocarbon groups (tails) (Figure 1). They are also known as surfactants because they decrease the surface tension of water.
How are detergents classified?
Detergents. Detergents are classified according to their chemistry when in solution: nonionic, anionic, or cationic. The nonionic and anionic detergents are of low toxicity although they may be mildly to moderately irritant. Most serious toxicoses are associated with the cationic detergents.
Why are detergents amphipathic molecules?
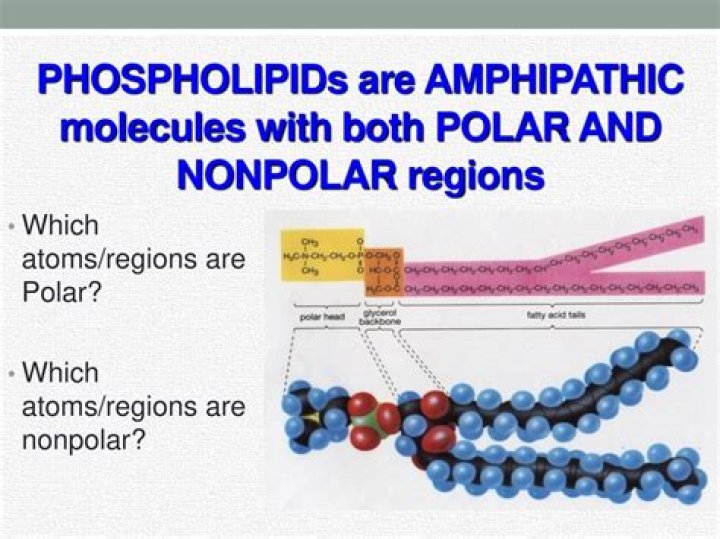
Detergents are amphipathic molecules that contain both polar and hydrophobic groups. These molecules contain a polar group (head) at the end of a long hydrophobic carbon chain (tail). In contrast to purely polar or non-polar molecules, amphipathic molecules exhibit unique properties in water.
Are soaps hydrophobic?
Soap is made of pin-shaped molecules, each of which has a hydrophilic head — it readily bonds with water — and a hydrophobic tail, which shuns water and prefers to link up with oils and fats.
Is detergent homogeneous or heterogeneous?
Examples Of Homogenous Mixtures Homogenous mixtures include mixtures such as laundry detergent, blood plasma, vinegar, and coffee.
How do detergents work chemistry?
How do detergents work? Soaps and detergents are made from long molecules that contain a head and tail. These molecules are called surfactants; the diagram below represents a surfactant molecule. The head of the molecule is attracted to water (hydrophilic) and the tail is attracted to grease and dirt (hydrophobic).
Does detergent soap absorb water?
Surfactants such as dish soap break up water’s surface tension. As a result, objects floating in water will sink or change shape as the surface tension changes.
Is oil and water polar or nonpolar?
Water molecules are polar molecules. That means one end of the molecule has a positive charge and the other end has a negative charge. This allows water molecules to bond together. Oil molecules, on the other hand, are non-polar.
Do amphipathic molecules have polar and nonpolar regions?
Amphipathic molecules have both polar and nonpolar regions. Dr. Helmenstine holds a Ph.D. in biomedical sciences and is a science writer, educator, and consultant. She has taught science courses at the high school, college, and graduate levels.
What are amphipathic molecules in soap?
Amphipathic molecules allow detergents, soaps, shampoos, and many other cleaning products to carry away substances that don’t wash away with water alone. Soaps are traditionally made by treating fatty substances, such as vegetable oils or animal fat, with a chemical called lye.
What is the difference between polar and nonpolar molecules?
Non-polar molecules, on the other hand, do not interact strongly with polar molecules and may actually be pushed out of the way by other polar molecules that are attracted to the polar molecules’ partial charges. Amphipathic molecules are biologically useful because they can interact with both polar and non-polar substances.
What is the difference between amphipathic molecules and micelle?
In aqueous solutions, amphipathic molecules spontaneously assemble into micelles. A micelle has lower free energy than free-floating amphipaths. The polar portion of the amphipath (the hydrophilic part) forms the outer surface of the micelle and is exposed to water.



