Is enthalpy exothermic or endothermic?
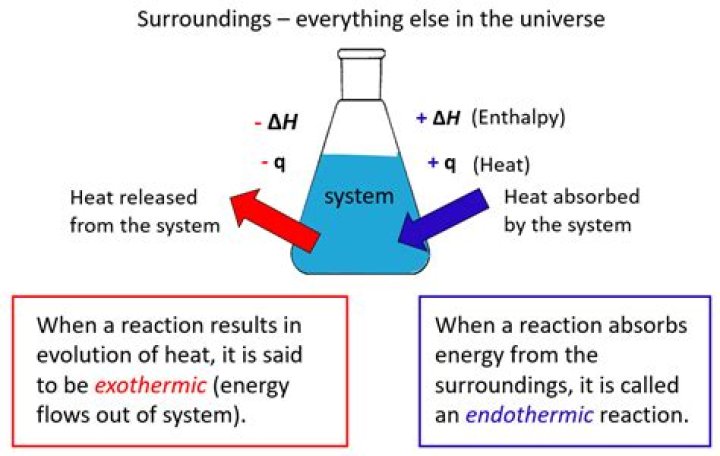
So, if a reaction releases more energy than it absorbs, the reaction is exothermic and enthalpy will be negative. Think of this as an amount of heat leaving (or being subtracted from) the reaction. If a reaction absorbs or uses more energy than it releases, the reaction is endothermic, and enthalpy will be positive.
Is gas production exothermic or endothermic?
Our experience makes it easy for us to realize that to boil water or any liquid and thereby convert into a gas, heat is required and the process is endothermic. It is less intuitive to grasp that when a gas condenses to a liquid, heat is given off and the process is exothermic.
Is enthalpy of a reaction exothermic?
The overall enthalpy of the reaction is negative, i.e., it’s an exothermic reaction where energy is released in the form of heat.
How do you know if its exothermic or endothermic?
If the enthalpy change listed for a reaction is negative, then that reaction releases heat as it proceeds — the reaction is exothermic (exo- = out). If the enthalpy change listed for the reaction is positive, then that reaction absorbs heat as it proceeds — the reaction is endothermic (endo- = in).
How does the enthalpy of the products compare with the enthalpy of the reactants?
A system of reactants that absorbs heat from the surroundings in an endothermic reaction has a positive ΔH, because the enthalpy of the products is higher than the enthalpy of the reactants of the system.
What is the enthalpy change for an endothermic reaction?
A reaction that absorbs energy is an endothermic reaction; its enthalpy change ( H) is positive. The enthalpy of the products of the reaction is greater than that of the reactants. Energy is absorbed from the surroundings.
Is Delta H negative for exothermic?
A system that releases heat to the surroundings, an exothermic reaction, has a negative ΔH by convention, because the enthalpy of the products is lower than the enthalpy of the reactants of the system. The enthalpies of these reactions are less than zero, and are therefore exothermic reactions.
What is the enthalpy of the products in an exothermic reaction?
In this type of enthalpy change, the enthalpy of the products is less than the enthalpy of the reactants. The value of the enthalpy change is negative (ΔH = -ve). The reaction between petrol and oxygen which I have mentioned is an example of an exothermic reaction. Because it releases heat or heats up the surrounding.
What is endendothermic and standard enthalpy?
endothermic: net absorbance of heat enthalpy: in thermodynamics, a measure of the heat content of a chemical or physical system standard enthalpy of formation: the change in enthalpy that accompanies the formation of one mole of a compound from its elements, with all substances in their standard states
How do you calculate the enthalpy of a reaction using Hess law?
Hess’s law can be used to calculate the enthalpy of reaction using the sum of the standard enthalpies of the intermediate reactions into which the overall reaction can be divided, while each occurs at the same temperature.
What is an endothermic reaction?
Endothermic Reaction- A reaction that causes the temperature of the surroundings to decrease. Energy is used up in the reaction, as the enthalpy of the products is greater than the enthalpy of the reactants. Most endothermic reactions are not spontaneous



