Is HClO a strong acid or base?
Hypochlorous acid (HOCl or HClO) is a weak acid that forms when chlorine dissolves in water, and itself partially dissociates, forming hypochlorite, ClO−.
Why is HClO a strong acid?
These acids are the only ones that fully dissociate in water. All other acids are weak acids. Since HClO is not one of these seven, and there is no -OH group present as there is in bases, HClO is a weak acid. Another way to verify that this is an acid instead of a base is by looking at the ion it dissociates into.
Is HClO an acid base or salt?
Classifying Electrolytes
| Strong Electrolytes | strong acids | HCl, HBr, HI, HNO3, HClO3, HClO4, and H2SO4 |
|---|---|---|
| strong bases | NaOH, KOH, LiOH, Ba(OH)2, and Ca(OH)2 | |
| salts | NaCl, KBr, MgCl2, and many, many more | |
| Weak Electrolytes | ||
| weak acids | HF, HC2H3O2 (acetic acid), H2CO3 (carbonic acid), H3PO4 (phosphoric acid), and many more |
What defines a strong acid or base?
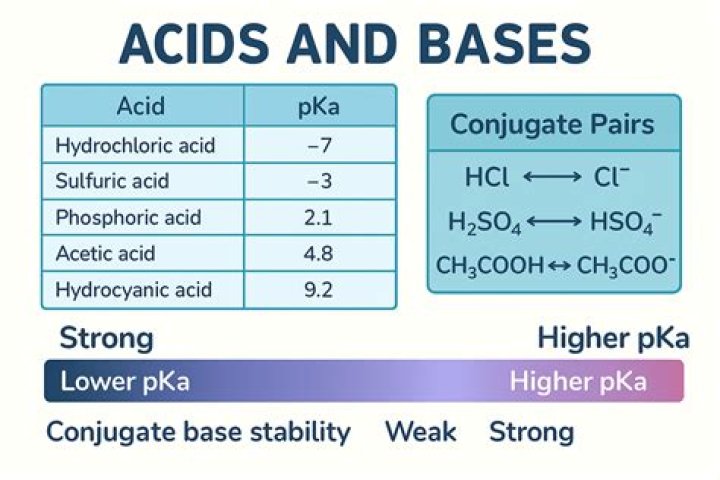
Strong acids are defined by their pKa. The acid must be stronger in aqueous solution than a hydronium ion, so its pKa must be lower than that of a hydronium ion. Therefore, strong acids have a pKa of <-174. Strong acids must be handled carefully because they can cause severe chemical burns.
Is HClO a weak or strong electrolyte?
HBr is a strong electrolyte. HClO is a weak electrolyte.
Is HClO a Bronsted base?
HClO4 act as Bronsted-Lowry acid as it donates the proton and forms a conjugate base. NH3 acts as a Bronsted-Lowry base as it accepts the proton from HClO4 and forms a Conjugate acid.
Is HClO a stronger acid than HCl?
In direct contrast with HCl vs. HBr , HClO is a stronger acid than HBrO , because Cl is more electronegative, which dominates over the size difference between Cl and Br due to the presence of the oxygen.
What makes a strong base?
A strong base is a base that is completely dissociated in an aqueous solution. These compounds ionize in water to yield one or more hydroxide ion (OH-) per molecule of base. Strong bases react with strong acids to form stable compounds.
How weak is HClO?
Hypochlorous acid, HClO, is a weak acid with Ka = 3.0 x 10-8.
Is HClO a strong electrolyte?
Problem: HBr is a strong electrolyte. HClO is a weak electrolyte. KClO(aq) + HBr(aq) → KBr(aq) + HClO(aq). Write the net ionic equation.
Is H2CO3 a strong acid?
Is carbonic acid a strong acid? No, carbonic acid is not a strong acid. H2CO3 is a weak acid that dissociates into a proton (H+ cation) and a bicarbonate ion (HCO3- anion). This compound only partly dissociates in aqueous solutions.
Is HClO is the strongest acid?
HClO is an acid as is has the proton that it can donate but it is a weak acid because it is not one the acid amoung the list of the strong acids.
Why is HCl a stronger acid than acetic acid?
EXPERT ANSWER. HCl dissociates completely in water and thus gives more H + ions whereas acetic acid dissociates partially in water and thus gives less H + ions. Due to this, HCl is a strong acid than acetic acid.
Why is HClO3 considered a strong acid?
2) HClO3 is stronger because chlorine is more electronegative than iodine. The more electronegative the element is the more strong or acidic it becomes. Chlorine being more electronegative than Iodine makes it easier for it to pull the electron of hydrogen more strongly and hence has a higher tendency to release a H + unit.
Is HCl is a strong base or acid?
They are part of the epithelial fundic glands (also known as oxyntic glands) in the stomach. The chemical compound hydrochloric acid is the aqueous (water-based) solution of hydrogen chloride gas (HCl). It is a strong acid, the major component of gastric acid and of wide industrial use.



