Is ICl3 ionic or molecular?
ICl3 is made from only nonmetals and is a covalent compound.
Does ICl3 have a resonance structure?
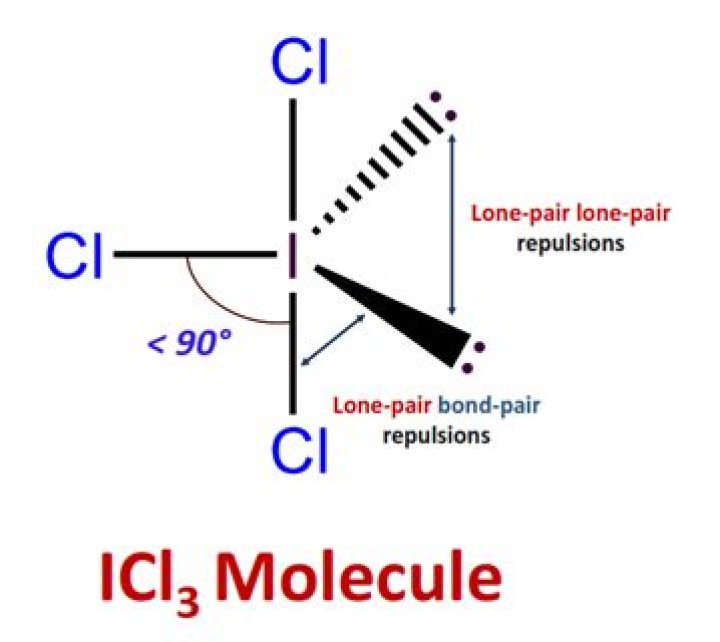
Iodine trichloride is also a T shaped molecule with trigonal bipyramidal structure. It contains three bond pairs and two lone pair of electrons. Both the molecules do not have resonance structures as bond and lone pairs of electrons are not involved in conjugation.
Is I3 linear?
I3- molecular geometry is linear. While there are three Iodine atoms, one of the atoms has a negative charge which further gives 3 lone pairs of electrons and 2 bond pairs. The three lone pairs will repel each other and take up equatorial positions.
Why is ICl3 not trigonal planar?
Re: Why Can’t T-shaped Molecules be Trigonal Planar? The reason why ICl3 exists in T-shaped geometry, with all of its angles at slightly less than 90 degrees, is because it is the most stable in that structure.
What is the shape of ICl3?
Geometry of ICl3 is tetrahedral.
Why I3 is non polar?
The bond angle between the two is 180 degrees and all three iodine atoms share electrons evenly. Thus, it has no net dipole moment. As DavidThomas noted in his answer, triiodide is never polar in the sense of having a slight positive and negative end.
Why is n3 linear?
Count the number of valence electrons of nitrogen atoms. The outer valence shell electronic configuration of N is $2{s^2}2{p^3}$. Therefore, valence electrons of three nitrogen atoms are 5+5+5 = 15. Thus, the structure of $N_3^ – $ molecule is linear in shape.
What is the correct molecular shape of ICl3?
The ICl3 molecule has a trigonal geometry shape because it contains three chlorine atoms. There are three I-Cl bonds at the ICl3 molecular geometry. After linking the three chlorine atoms and two lone pairs of electrons in the trigonal bipyramidal form, it maintains the distorted T-shaped structure.
Why is ICl3 trigonal planar?
The reason why ICl3 exists in T-shaped geometry, with all of its angles at slightly less than 90 degrees, is because it is the most stable in that structure.
Is ICl3 tetrahedral?
Is ClO3 -1 polar?
With a non-zero dipole moment, the ClO3- ion is a polar molecule . Due to its asymmetrical shape and the presence of a net dipole moment, the ClO3- ion is polar in nature. This affects its solubility and other properties.
Is i3- polar or nonpolar?
So it is neither polar or nonpolar. However, if you have to describe the ion, you can use the phrase the “ like a polar molecule” because I3- is soluble in water. In concluding remarks, to sum up, this entire article, I3- is a polyatomic ion that has 22 valence electrons, 3 lone pairs, 2 bond pairs, and sp3d hybridization.
Is ICL polar non polar or ionic?
ICl is polar and Br2 is nonpolar. The dominate intermolecular force in ICl is dipole- dipole whereas in Br2 it is London. Dipole-dipole is the stronger of the two and therefore more difficult to break – the melting point is higher.
What is the molecular geometry of ICl3?
The Lewis structure of ICl3 is a drawing or model chemists use to predict the geometry of the molecule. ICl3 is one of the compounds that don’t follow the octet rule, as the iodine at the center of this model accepts 10 electrons. Each chlorine atom in the molecule provides seven valence electrons, for a total of 21.



