Is nitrogen-14 used in carbon dating?
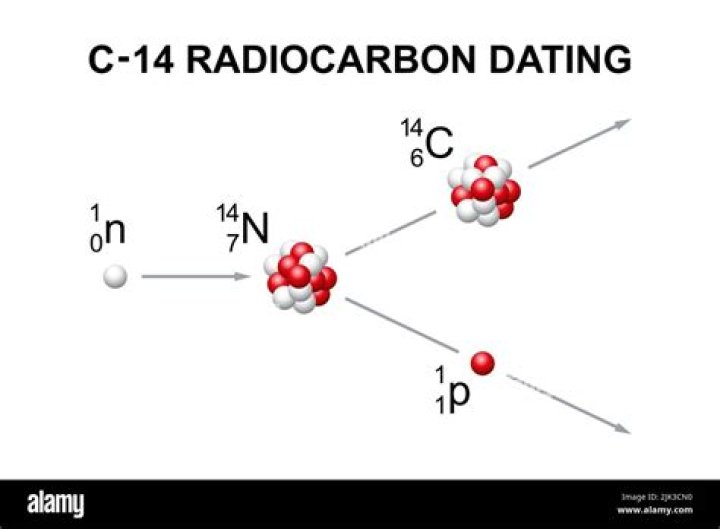
Carbon-14 is continually formed in nature by the interaction of neutrons with nitrogen-14 in the Earth’s atmosphere; the neutrons required for this reaction are produced by cosmic rays interacting with the atmosphere. …
What is the relationship between nitrogen and carbon-14?
The relationship between carbon-14 and nitrogen is that carbon-14 forms from nitrogen atoms and eventual decays into atoms of nitrogen-14.
How does nitrogen-14 become carbon-14?
How Carbon-14 is Made. When the neutron collides, a nitrogen-14 (seven protons, seven neutrons) atom turns into a carbon-14 atom (six protons, eight neutrons) and a hydrogen atom (one proton, zero neutrons). Carbon-14 is radioactive, with a half-life of about 5,700 years.
Are carbon-14 and nitrogen 14 isotopes?
Carbon-12 and carbon-13 are both stable, while carbon-14 is unstable and has a half-life of 5,730 ± 40 years. Carbon-14 decays into nitrogen-14 through beta decay….Carbon-14.
| General | |
|---|---|
| Beta | 0.156476 |
| Isotopes of carbon Complete table of nuclides |
Is carbon-14 an isotope of carbon?
carbon-14, the longest-lived radioactive isotope of carbon, whose decay allows the accurate dating of archaeological artifacts. The carbon-14 nucleus has six protons and eight neutrons, for an atomic mass of 14.
How is carbon 14 dating done?
Radiocarbon dating works by comparing the three different isotopes of carbon. Isotopes of a particular element have the same number of protons in their nucleus, but different numbers of neutrons. This means its nucleus is so large that it is unstable. Over time 14C decays to nitrogen (14N).
Are carbon-14 and nitrogen-14 isotopes of each other?
They all have the same atomic number, same number of protons. Explain why carbon-14 and nitrogen-14 are not considered isotopes of each other? Because they are two different elements. Same mass number but different atomic number.
For which example is carbon 14 dating appropriate for dating?
Archaeology and other human sciences use radiocarbon dating to prove or disprove theories. Over the years, carbon 14 dating has also found applications in geology, hydrology, geophysics, atmospheric science, oceanography, paleoclimatology and even biomedicine.
Whats the difference between carbon-14 and nitrogen-14?
Having different atomic number means that they have different properties. Carbon 14 forms 4 bonds and nitrogen forms 3 bonds. carbon 14 is a radioactive isotope of carbon because it has more neutrons than protons. nitrogen 14 is not radioactive.
Is nitrogen-14 an isotope?
Nitrogen-14 is one of two stable (non-radioactive) isotopes of the chemical element nitrogen, which makes about 99.636% of natural nitrogen. Nitrogen-14 is one of the very few stable nuclides with both an odd number of protons and of neutrons (seven each) and is the only one to make up a majority of its element.
How is the isotope carbon dating done?
Carbon dating is a dating technique predicated upon three things: 1 The rate at which the unstable radioactive C-14 isotope decays into the stable non-radioactive N-14 isotope, 2 The ratio of C-12 to C-14 found in a given specimen, 3 And the ratio C-12 to C-14 found in the atmosphere at the time of the specimen’s death. More
What is the age of carbon 14 dating?
Carbon-14 dating can be used to estimate the age of carbon-bearing materials up to about 58,000 to 62,000 years old. The carbon-14 isotope would vanish from Earth’s atmosphere in less than a million years were it not for the constant influx of cosmic rays interacting with atmospheric nitrogen.
What are the basic principles of carbon dating?
Basic Principles of Carbon Dating. Radiocarbon, or carbon 14, is an isotope of the element carbon that is unstable and weakly radioactive. The stable isotopes are carbon 12 and carbon 13. Carbon 14 is continually being formed in the upper atmosphere by the effect of cosmic ray neutrons on nitrogen 14 atoms.
Why is the isotope 14 C important?
Although 12 C is definitely essential to life, its unstable sister isotope 14 C has become of extreme importance to the science world. Radiocarbon dating is the process of determining the age of a sample by examining the amount of 14 C remaining against its known half-life, 5,730 years.



