Is paraffin a long-chain hydrocarbon?
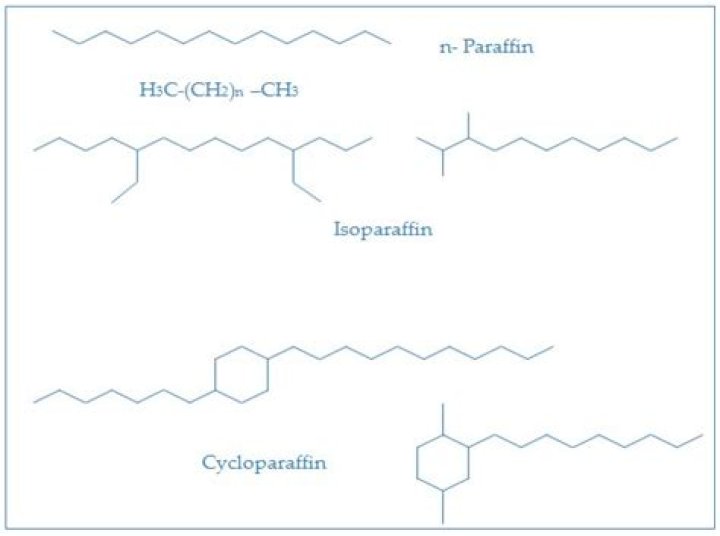
Paraffins are straight- or branched-chain hydrocarbons having the chemical formula CnH2n+2.
How many carbon atoms does paraffin have in its chain?
Paraffin wax (or petroleum wax) is a soft colorless solid derived from petroleum, coal or oil shale that consists of a mixture of hydrocarbon molecules containing between twenty and forty carbon atoms.
How long is beeswax wax hydrocarbon chain?
The esters consist of long-chain fatty alcohols (C24-C38) and fatty acids, mainly palmitic acid (C16) and oleic acid (C18). Monoesters with a total chain length of 40-48 carbon atoms are quantitatively the most important, but C38, C50, C52 and C54 monoesters are also present.
How many carbons are in wax?
A wax is a simple lipid which is an ester of a long-chain alcohol and a fatty acid. The alcohol may contain from 12-32 carbon atoms. Waxes are found in nature as coatings on leaves and stems.
Why is paraffin nonpolar?
Looking back at their structure, paraffin wax contains C-H bonds all over the structure which makes the entire molecule neutral or without partial charges. This accounts to their insolubility with water. Paraffin waxes are insoluble because of its non-polar characteristic. All alkanes are non-polar in nature.
What is wax paraffin?
Paraffin wax is a white or colorless soft, solid wax. It’s made from saturated hydrocarbons. It’s often used in skin-softening salon and spa treatments on the hands, cuticles, and feet because it’s colorless, tasteless, and odorless. It can also be used to provide pain relief to sore joints and muscles.
How long does it take to melt paraffin wax?
Place your bowl/jug of wax in the microwave and heat for 1 minute at a time, monitoring the temperature until it reaches 80°C. (Do not let you wax exceed 80°C). Continue heating your wax in 2-minute intervals until it’s fully melted.
Why is paraffin wax bad?
Most candles today are made of paraffin wax which creates highly toxic benzene and toluene when burned (both are known carcinogens). In fact, the toxins released from paraffin candles are the same as those found in diesel fuel fumes and are linked to asthma and lung cancer.
Is paraffin wax a lipid?
A wax is a simple lipid that is an ester of a long-chain alcohol and a fatty acid. Paraffin wax which is used in some candles, is not based upon the ester functional group, but instead is a mixture of high molecular weight alkanes.
What is the percentage of carbon in paraffin?
The paraffinic carbon content is typically between 50 wt% and 65 wt% of the total feed. Paraffinic stocks are easy to crack and normally yield the greatest amount of total liquid products. Normal paraffins will crack mostly to olefin and other paraffin molecules.
What is paraffin wax formula?
The general formula for paraffin wax involves n carbon atoms and 2n plus 2 hydrogen atoms, where n is at least 16. For example, one of the hydrocarbons in the wax might have the formula C31H64. Manufacturers make the wax by removing the oil from a waxy byproduct of petroleum distillation.
Why paraffin wax does not dissolve in water?
If you put a lump of paraffin wax, which consists of many carbon and hydrogen bonds, into water, it remains as a lump. Even if you crush the wax up into little pieces and stir it around in the water, it still won’t dissolve. This is because water is polar and the wax is non-polar.
How do paraffin waxes differ from each other?
The results show that paraffin waxes differ significantly in composition and that heterogeneities within samples can arise when long chain n -alkanes become redistributed, while wax is cooling from a fluid to a solid.
How does paraffin wax cause corrosion in heat exchangers?
Paraffin waxes that stick to piping and vessel walls can trap amines against the walls, which can create localized corrosion. The severe fouling in heat exchangers upstream of the desalter was initially a surprise to many refiners – fouling is typically worse in the hotter exchangers that are downstream of the desalter.
How to remove paraffin Clearant from wax?
Due to the viscosity of molten paraffin wax some form of gentle agitation is highly desirable. It serves to remove the clearant from around the tissue and permit the access of uncontaminated wax.
How do you dissolve wax in an oil pipeline?
Wax, paraffin, or sulfur deposits in an oil or gas pipeline can be dissolved by addition of a compound which liberates carbon disulfide on contact with acid. Thereby the deposit is dissolved by the liberated carbon disulfide [ 20 ].



