Is PF5 sp2 hybridized?
there are 5 sigma bonds in this compound. hence , the hybridization of this compound is sp3d . 2 has sp hybridization. 3 has sp2 hybridization.
What are the shape of hybridisation of PF5?
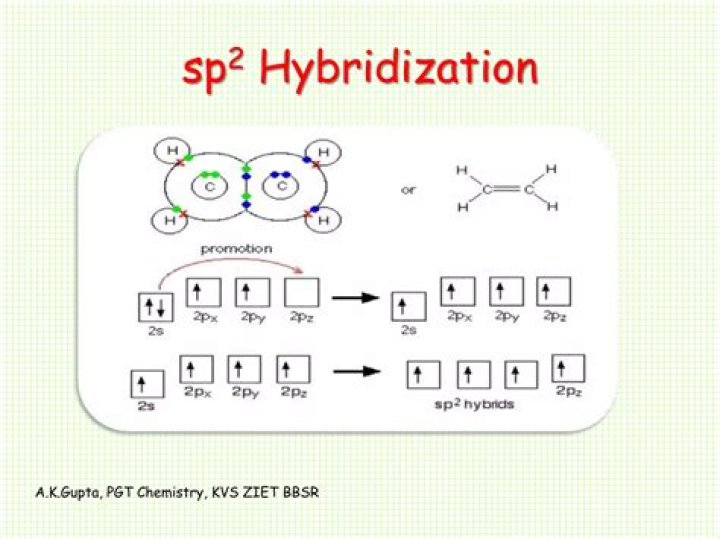
Thus PF5 has trigonal bipyramidal shape. Discuss in brief sp2 hybridization (hybridization in C = C bond).
What is geometry of PF5?
The molecular geometry of PF5 is trigonal bipyramidal with symmetric charge distribution.
What is the shape of PF5 according to Vsepr theory?
Thus it forms five bonds with fluorine atoms and no lone pairs present on it….Note:
| Total number of electron pairs | Shape |
|---|---|
| 7 | Pentagonal bipyramidal |
What is the hybridization of the central atom of each of the following molecules PF5?
The hybridization of the central atom, P, is sp3d because it’s shape is trigonal bipyramidal.
What type of hybridized Orbital can be seen in carbon tetrachloride?
Pz orbital = 1 Valence electron pair. This takes us to the conclusion that the hybridization of CCl4 is sp3.
What’s the structure of PF5 Is it polar or nonpolar?
Phosphorus pentafluoride or PF5 is a nonpolar compound because of its symmetrical geometry ie; Trigonal Bipyramidal.
What is the bond angle of PF5?
The resulting shape is a trigonal bipyramidal in which three fluorine atoms occupy equatorial and two occupy axial positions. The F-P-F angle between equatorial positions is 120°, between the axial and equatorial positions it is 90°.
What is the point group of PF5?
The PF5 molecule belongs to the D3h point group.
What is the hybridization of the central atom in the beryllium chloride molecule?
sp hybridization
During the formation of BeCl2, beryllium atom bonds with two chlorine atoms via single covalent bonds. The number of electron pairs around the central atom will be two. No lone pair is found in the molecule. If we analyse this information then we can conclude that BeCl2 has sp hybridization.
Why is beh2 sp hybridized?
A beryllium hydride (BeH2) molecule is predicted to be linear by VSEPR. The beryllium atom contains only paired electrons, so it must also undergo hybridization. One of the 2s electrons is first promoted to an empty 2p orbital. The occupied orbitals are then hybridized, and the result is a pair of sp hybrid orbitals.
What is the electron geometry of PF5?
The molecular geometry of PF5, phosphorus pentafluoride, is triangular bipyramidal. It has 10 electrons surrounding a central phosphorous atom, creating five electron pairs in a trigonal bipyramidal shape.
The PF5 bond angle will be about 120 and 90 degrees (see video for where 90 and 120 degree bond angles are found on PF5) since it has a trigonal bipyramidal molecular geometry. Get more chemistry help at
What are the shapes of PF5?
The basic geometric shape of a molecule containing a central atom with three pairs of electrons is a plane triangle . The molecular geometry of PF5 is triangular bipyramidal, and electronic geometry of PF5 is the plane triangle.
How to determine hybridization?
If it’s 4,your atom is sp 3.



