Is vanadium sulfide ionic or covalent?
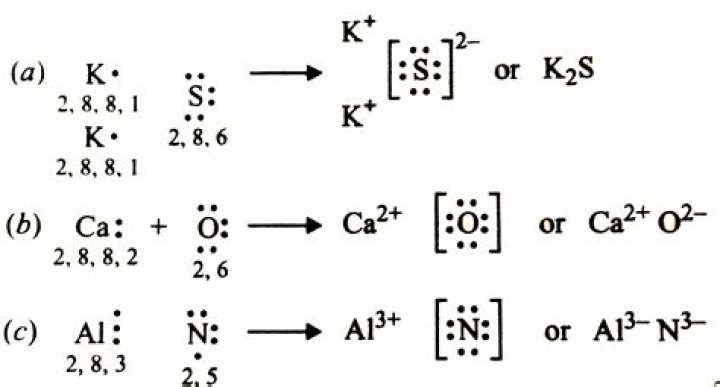
This compound is ionic and thus we follow naming as follow: 1. Vanadium, V, is an element with multiple charges.
How do you know if it is a covalent or ionic bond?
Classifying compounds as ionic or covalent
- If a compound is made from a metal and a non-metal, its bonding will be ionic.
- If a compound is made from two non-metals, its bonding will be covalent.
Does VF5 exist?
Vanadium(V) fluoride is the inorganic compound with the chemical formula VF5. It is a colorless volatile liquid.
What is the structure of vanadium Pentafluoride?
The structure of vanadium pentafluoride, VF5. VF5 has a structure that consists of an infinite chain of molecules linked by cis-bridging fluorides. The vanadium(V) is approximately octahedral. The white compound has a melting point of 19.5°C and a boiling point of 48.3°C.
What compound is Ni2S3?
Nickel sulfide
Nickel sulfide (Ni2S3)
| PubChem CID | 183144 |
|---|---|
| Synonyms | Nickel sulfide (Ni2S3) 12259-56-2 Nickel(3 ) sulfide DTXSID10153657 |
| Molecular Weight | 219.6 |
| Component Compounds | CID 402 (Hydrogen sulfide) CID 935 (Nickel) |
| Dates | Modify 2021-11-06 Create 2005-08-09 |
What is a vf5?
Vanadium(V) fluoride, a chemical compound.
Why vanadium does not form vcl5?
Vanadium exhibits highest oxidation state in. With iodine vanadium cannot form VI5 due to. oxidation power of V5+ and reducing nature of I.
What is the chemical formula of vanadium III selenide?
Vanadium(III) Selenide V2Se3 Molecular Weight — EndMemo.
What is the formula of beryllium oxide?
BeO
Beryllium oxide/Formula
What is the difference between ionic and covalent bonding?
Ionic and Covalent Bonding There are primarily two forms of bonding that an atom can participate in: Covalent and Ionic. Covalent bonding involves the sharing of electrons between two or more atoms. Ionic bonds form when two or more ions come together and are held together by charge differences.
Can a covalent bond form between two nonmetals?
The only pure covalent bonds occur between identical atoms. Usually, there is some polarity (polar covalent bond) in which the electrons are shared, but spend more time with one atom than the other. Ionic bonds form between a metal and a nonmetal. Covalent bonds form between two nonmetals.
How can you tell if a compound is a covalent bond?
Usually, an electron is more attracted to one atom than to another, forming a polar covalent bond. For example, the atoms in water, H 2 O, are held together by polar covalent bonds. You can predict a covalent bond will form between two nonmetallic atoms. Also, covalent compounds may dissolve in water, but don’t dissociate into ions.
What are the rules for building a covalent bond?
There are a couple of rules to follow however as you build: The Octet Rule: The atoms that participate in covalent bonding share electrons in a way that enables them to acquire a stable electron configuration, or full valence shell. This means that they want to acquire the electronic configuration of the noble gas of their row.



