What are donor and acceptor impurities?
Donor vs Acceptor Impurities Donor impurities are the elements added to a donor to increase the electrical conductivity of that donor. Acceptor impurities are the elements added to an acceptor to increase the electrical conductivity of that acceptor.
What are the acceptor impurities in semiconductor?
Antimony, Phosphorus and Arsenic have 5 valence electrons hence they act as donor impurity. Indium has 3 valence electrons and hence it is an acceptor impurity.
What are the acceptor impurities?
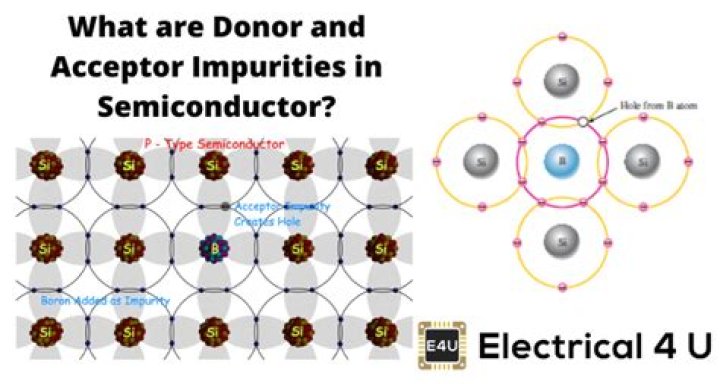
An acceptor Impurity is a physical material which when added to a semiconductor can form P-type region by creating positive charges or holes in the semiconductor material like silicon or germanium.
What is donor and acceptor level in semiconductor?
Donors are impurities which contribute levels that are just below the conduction band edge. Acceptors are impurities which have less valence electrons than the host, e.g., gallium (valence=3) doped into germanium (valence=4). Acceptors contribute impurity energy levels just above the valence band edge.
What is donor acceptor?
Donor / Acceptor Defined A donor is a high energy orbital with one or more electrons. An acceptor is a low energy orbital with one or more vacancies: A donor is an atom or group of atoms whose highest filled atomic orbital or molecular orbital is higher in energy than that of a reference orbital.
Which type of impurity is called acceptor impurity?
Due to the presence of less number of electrons in the valence shell however, group III elements of the periodic table are considered as acceptor impurity. Note: When doped with a semiconductor, if a dopant has 3 electrons in its valence shell to raise its conductivity, it is known as an acceptor impurity.
Is p-type donor or acceptor?
Because an acceptor donates excess holes, which are considered to be positively charged, a semiconductor that has been doped with an acceptor is called a p-type semiconductor; “p” stands for positive.
What is a donor and acceptor?
Which of the following is an example of electron acceptor impurities in semiconductors?
If an acceptor impurity is added to a semiconductor, it can also be a dopant atom which can form a p-type region. These atoms have electrons but four in their outermost shell, and hence they accept electrons from nearby atoms. The examples of such atoms are boron and aluminium.
What is donor acceptor mechanism?
Description. The donor-acceptor interaction could be considered as charge transfer between the donor and acceptor molecules without the formation of a chemical bond between them and the transfer of a lone pair of electrons from the donor to the acceptor, resulting in the formation of a bond.
What is donor and acceptor ion?
Electron acceptors are ions or molecules that act as oxidizing agents in chemical reactions. Electron donors are ions or molecules that donate electrons and are reducing agents. In the combustion reaction of gaseous hydrogen and oxygen to produce water (H2O), two hydrogen atoms donate their electrons to an oxygen atom.
What are donor and acceptor impurities in semiconductor materials?
Elements like phosphorus, antimony, bismuth, arsenic etc. are donor impurities. While boron, gallium, aluminium etc. are acceptor impurity atoms. So, from this discussion, we can conclude that impurity is added in order to enhance the conductivity of the semiconductor.
Which impurity is used to form n-type semiconductors?
Thus donor impurity is used to form n-type semiconductors. Group V elements are said to be donor impurity because they consist of 5 electrons in the outermost shell. Thus it is also known as pentavalent impurity. Consider that a pentavalent impurity Arsenic (As) is doped into a pure silicon structure.
What is the Acceptor impurity of a dopant?
A dopant with 3 electrons in its valence shell, when doped with a semiconductor to raise its conductivity, is known as an acceptor impurity. It has the ability by which it can accept an electron from neighbouring atom as it has a vacancy of electron. Thus is called acceptor impurity.
Which of the following elements is a Donor impurity?
Group III elements are known as donor impurity because these elements consist of 3 electrons in the valence shell. Thus is known as trivalent impurity. Elements like boron, aluminium, indium and gallium are examples of trivalent impurity. Consider an aluminium atom is doped in a pure crystal of silicon:



