What are energy band in solids?
In a single isolated atom, the electrons in each orbit have definite energy associated with it. The electrons in same orbit exhibits different energy levels. The grouping of this different energy levels is called energy band.
What is band structure of solids?
In solid-state physics, the electronic band structure (or simply band structure) of a solid describes the range of energy levels that electrons may have within it, as well as the ranges of energy that they may not have (called band gaps or forbidden bands).
What are energy bands in solids how are they formed?
In molecules, Two atomic orbitals combine together to form a molecular orbit with two distinct energy levels. In solids, 1023 stacked up lines confined in a tiny space would look like a band. Thereby forming energy continuum called energy bands.
What are energy bands and energy levels in solids?
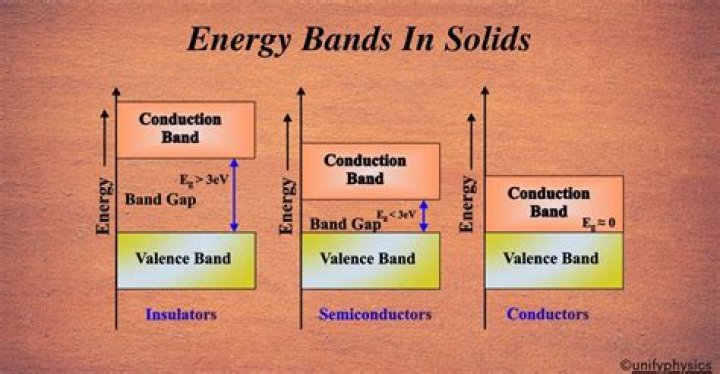
The valence band comprises the highest energy electrons in the solid, and the conduction band is the lowest empty belt where electrons can remain. These bands are the permissible bands. The energy band between the permissible bands is the band gap or forbidden band, where the electrons cannot exist.
What are energy bands?
Energy band theory is a theory of a valence electron moving in a periodic potential field of a crystalline lattice. Single atoms have a discrete energy spectrum, which means they can occupy only discrete energy levels. Part of these energy levels are filled with electrons in a non-excited condition.
What is energy band explain?
Which are the important energy bands in solids?
Valence Bands, Conduction bands and forbidden bands are the three important energy bands in solids.
What is energy band and its types?
Energy band theory explains the interaction of electrons between the outermost shell and the innermost shell. Based on the energy band theory, there are three different energy bands: Valence band. Forbidden energy gap. Conduction band.
What is energy band structure in solid?
Energy Band Structures In Solids. When atoms come together to form a solid, their valence electrons interact due to Coulomb forces, and they also feel the electric field produced by their own nucleus and that of the other atoms.
How is the band theory of solids different from the others?
The band theory of solids is different from the others because the atoms are arranged very close to each other such that the energy levels of the outermost orbital electrons are affected. But the energy level of the innermost electrons is not affected by the neighboring atoms.
What is band theory in quantum physics?
Band Theory was developed from the knowledge gained during the quantum revolution in science. In 1928, Felix Bloch applied quantum theory to solids. In atoms, electrons are filled in respective energy orbits following Pauli’s exclusion principle. Two atomic orbitals combine to form a molecular orbit with two distinct energy levels.
Which energy band is present below the conduction band?
The valence band is present below the conduction band and the electrons of this band are loosely bound to the nucleus of the atom. The energy band that consists of free electrons energy levels, is known as the conduction band.



