What are examples of hypotonic IV solutions?
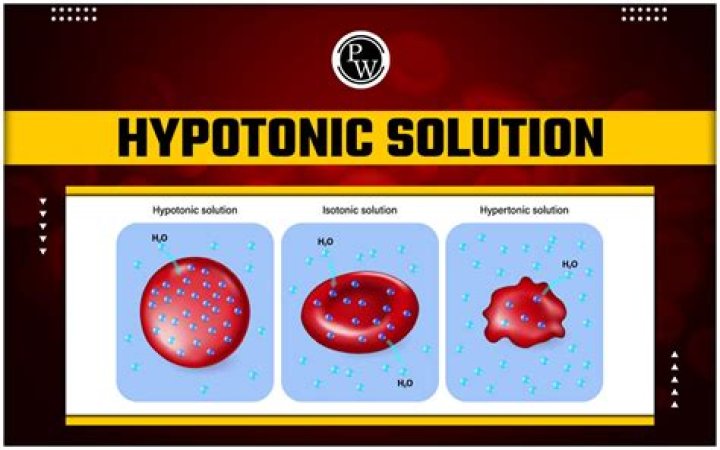
Cells will swell but may also delete fluid within the vascular space. Examples of hypotonic solutions include 0.45% sodium chloride, 0.33% sodium chloride, 2.5% dextrose in water, and 0.2% sodium chloride.
What are the examples of isotonic hypertonic and hypotonic IV solution?
Hypertonic, Hypotonic, Isotonic IV solutions
- Hypertonic: D5 NaCl. D5 in Lactated ringers. D5 0.45% NaCl.
- Isotonic: 0.9% NaCl (Normal Saline) Lactated Ringers. D5W (In the bag)
- Hypotonic: D5W (in the body) 0.25% NaCl. 0.45% NaCl (half normal saline) 2.5% Dextrose.
What is the tonicity of IV fluids?
The Infusion Nurses Society (INS) classifies a solution as isotonic if its tonicity falls within (or near) the normal range for blood serum-between 280 and 300 mOsm/liter. A hypotonic solution has an osmolarity less than 280 mOsm/liter, and a hypertonic solution has an osmolarity greater than 300 mOsm/liter.
What type of solution is used in IV fluids?
Normal saline solution can be administered only via intravenous (IV) access. 0.9% Normal Saline (NS, 0.9NaCl, or NSS) is one of the most common IV fluids, it is administered for most hydration needs: hemorrhage, vomiting, diarrhea, hemorrhage, drainage from GI suction, metabolic acidosis, or shock.
How is tonicity affected by solutes?
Tonicity describes how an extracellular solution can change the volume of a cell by affecting osmosis. A solution with low osmolarity has a greater number of water molecules relative to the number of solute particles; a solution with high osmolarity has fewer water molecules with respect to solute particles.
Which IV fluids for dehydration?
Isotonic: This is the most common type of IV fluid. Isotonic IV fluids include normal saline, 5% dextrose solutions dissolved in water, and Lactated Ringer’s solutions. These are used for dehydration caused by electrolyte imbalances as well as fluid loss from diarrhea and vomiting.
Why do intravenous IV solutions need to have the same tonicity as blood?
It is important for IV to be isotonic to your blood so it doesn’t change the concentration of different molecules in the bloodstream. the cell wall will start to shrink, due to loss of the fluid. High osmotic pressure with allow fluids to pull fluids out of the cell.
What is an example of an isotonic IV solution?
An example of an isotonic IV solution is 0.9% Normal Saline (0.9% NaCl). Because the concentration of the IV fluid is similar to the blood, the fluid stays in the intravascular space and osmosis does not cause fluid movement between compartments.
What is the Cheat Sheet for hypotonic IV fluids?
Cheat sheet for Hypotonic IV Fluids. 0.33% Sodium Chloride Solution is used to allow kidneys to retain the needed amounts of water and is typically administered with dextrose to increase tonicity. It should be used in caution for patients with heart failure and renal insufficiency. 0.225% Sodium Chloride (0.225% NaCl)
What are the different types of intravenous solutions?
Get to know the different types of intravenous solutions or IV fluids in this guide and cheat sheet. Differentiate isotonic, hypertonic, and hypotonic IV solutions and the nursing interventions and management for each. What are IV Fluids? What are IV Fluids?
What are the different types of IV fluids?
There are three types of IV fluids: isotonic, hypotonic, and hypertonic. are IV fluids that have a similar concentration of dissolved particles as blood. An example of an isotonic IV solution is 0.9% Normal Saline (0.9% NaCl).



