What are the approximate bond angles in XeCl2?
180° is the value of the bond angle in XeCl2.
What are the bond angles of H2O?
The bond angle in H2O is 104.50.
What is the molecular geometry of XeCl2?
Xenon dichloride – XeCl2 : The molecular geometry of XeCl2 is linear with symmetric electron region distribution.
Does H2O have a bond angle of 120?
The H-O-H bond angle in H2O is about 109°, whereas the bond angle for O-S-O is about 120°. Explain using lewis diagrams why the molecules have the same molecular geometry but different bond angles.
How many bonding electrons does XeCl2 have?
XeCl2 has 8 + 2(7) = 22 valence electrons. a linear molecular structure with a 180° bond angle. 7 + (4×7) -1 = 34 electrons.
What is the electron geometry of h2o?
For example; four electron pairs are distributed in a tetrahedral shape. If there are two bond pairs and two lone pairs of electrons the molecular geometry is angular or bent (e.g. H2O).
What is the shape and bond angle of H2O?
Water has 4 regions of electron density around the central oxygen atom (2 bonds and 2 lone pairs). These are arranged in a tetrahedral shape. The resulting molecular shape is bent with an H-O-H angle of 104.5°.
Is h20 linear or bent?
The water molecule is bent molecular geometry because the lone electron pairs, although still exerting influence on the shape, are invisible when looking at molecular geometry.
What is the molecular structure and bond angles for xecl2?
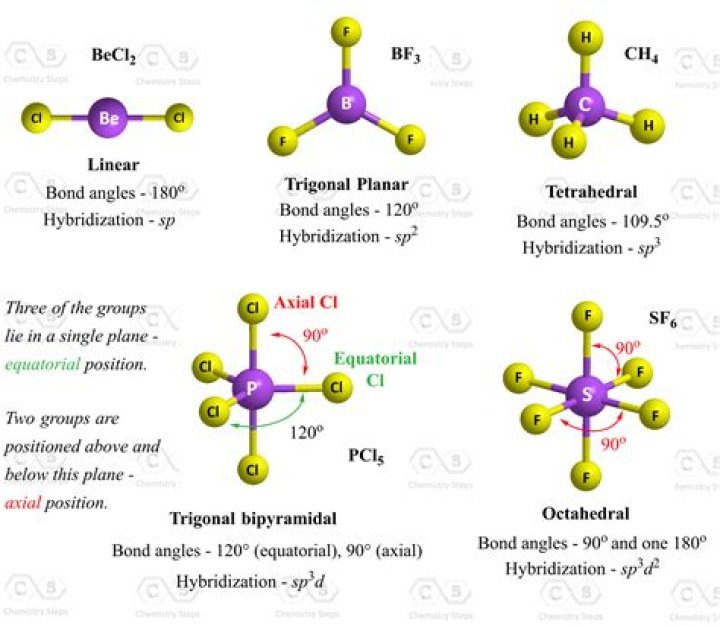
The complete Lewis structure or molecular structure of XeCl2 is: Step 4: Since there are 5 electron groups (2 atoms + 3 lone pairs) around Xe, the electron geometry is trigonal bipyramidal, whose ideal bond angles are 90˚, 120˚, and 180˚.
Why is the bond angle in water 105?
Because each water molecule has its hydrogen atoms about 105 degrees apart, and because these hydrogen atoms are attracted to the oxygen of other water molecules, a crystal begins to form. The water molecules within the crystal are attracted to other water molecules, but only at very precise angles.
What is bond angle of water?
The actual bond angle in the water molecule is 104.5°.
What is the value of the bond angle in XeCl2?
180° is the value of the bond angle in XeCl2. Explanation. The geometry of a covalent molecule can be predicted using valence shell electron pair repulsion theory (VSEPR theory). The Lewis dot structure of XeCl2 reveals three pairs of non-bonding, or lone electrons, and two pairs of bond electrons around Xe.
What are the properties of XeCl2?
Properties of XeCl2 1 Its molar mass is 202.199 g/mol and has a total of 22 valence electrons present. 2 It has a linear geometry where the bond angle between Xe-Cl bond is 180°. 3 XeCl2 hybridization is sp3d types and Xe oxidation state is +2.
What is the bond angle of a H2O molecule?
H2O, which is a three atom molecule, comes with the angular shape. H2O Bond Angles Looking at the table, when we go from AX2, AX3 and all the way down to AX2N2, we will find out that the bond angle is going to be 109.5 degrees.
What is the axial position of XeCl2?
In the case of XeCl2, there are three lone electron pairs around Xe that occupy equatorial positions, and therefore the two bond pairs (and hence the Cl atoms) occupy axial positions. The line passing through the Cl and Xe atoms is perpendicular to the equatorial plane containing the three lone electron pairs and the central atom Xe.



