What are the main types of bonds?
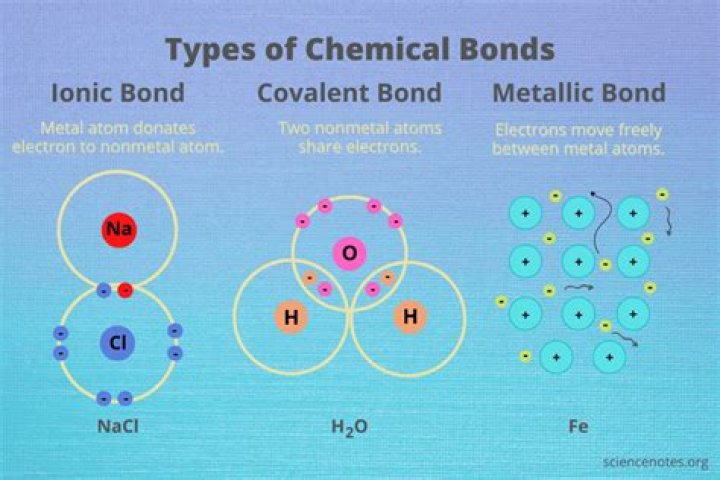
There are three primary types of bonding: ionic, covalent, and metallic.
- Ionic bonding.
- Covalent bonding.
- Metallic bonding.
What are the two strongest chemical bonds?
Covalent Bonds These bonds form when an electron is shared between two elements. Covalent bonds are the strongest (*see note below) and most common form of chemical bond in living organisms. The hydrogen and oxygen atoms that combine to form water molecules are bound together by strong covalent bonds.
What are the most common types of chemical bonds?
Covalent Bond The covalent bond is the strongest and most common form of chemical bond in living organisms. Together with the ionic bond, they form the two most important chemical bonds [1-7]. A covalent bond can be divided into a nonpolar covalent bond and a polar covalent bond.
What are 2 bonds called?
covalent bond
Let’s consider the covalent bond in the hydrogen molecule. A hydrogen molecule forms from two hydrogen atoms, each with one electron in a 1 s orbital. The two hydrogen atoms are attracted to the same pair of electrons in the covalent bond….Covalent Bonds.
| Atom | Valence |
|---|---|
| Nitrogen | 3 |
| Carbon | 4 |
What are the 4 types of chemical bonds?
There are four types of chemical bonds essential for life to exist: Ionic Bonds, Covalent Bonds, Hydrogen Bonds, and van der Waals interactions. We need all of these different kinds of bonds to play various roles in biochemical interactions. These bonds vary in their strengths.
What is chemical bonding and its types?
Chemical bonds are forces that hold atoms together to make compounds or molecules. Chemical bonds include covalent, polar covalent, and ionic bonds. Atoms with relatively similar electronegativities share electrons between them and are connected by covalent bonds. This attraction is known as an ionic bond.
What are the four main types of chemical bonds?
What are the 4 types of chemical bonding?
What are the three basic types of chemical bonds?
There are three main types of bonds: ionic, covalent and metallic. Ionic. These bonds occur when electrons are transferred from one atom two another, and are a result of the attraction between the resulting oppositely charged ions.
What are the three types of chemical bonding?
You can find three types of chemical bonds: Ionic, Covalent as well as Polar covalent. A COVALENT BOND results when two atoms “share” valence electrons between them. An IONIC BOND occurs when one atom gains the valence electron from the different atom, forming a negative ion (ANION) along with a positive ion (CATION), respectively.
What is the most common type of chemical bond?
Most common type of chemical bonding is single covalent bonding, where one pair of valence electrons is shared by the two atoms. Valence electrons are those that are in the outer orbit or shell of an atom. A good example of single covalent bonding is the Hydrogen molecule (H2).
What are the two ways that a chemical bond may form?
Chemical bonds form by the attraction the electrons of one atom to the proton of another atom. Common types of bonds include ionic, covalent and metallic bonds. The formation of the bond links the two atoms through the strong attractive forces, using a bond, a region where the electrons of the atoms interact with one another.



