What are the physical properties of sodium oxide?
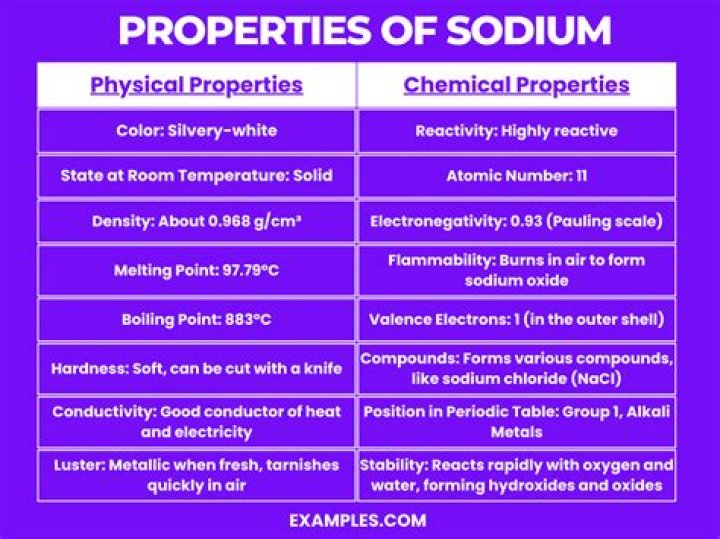
Physical properties: Sodium oxide is a white crystalline solid. Its melting and boiling points are 1132 °C and 1132 °C, respectively. Sodium oxide density is 2.27 g mL-1. It reacts violently with water.
Is sodium oxide hazardous?
Serious by all routes of exposure. The substance is corrosive to the eyes, skin and respiratory tract. Corrosive on ingestion. Inhalation of the aerosol may cause lung oedema.
What are sodium oxide used for?
Sodium oxide is a chemical compound with the formula Na2O. It is used in ceramics and glasses. The compound is the base anhydride of sodium hydroxide; when water is added to sodium oxide, NaOH is produced.
What is the form of sodium oxide?
Sodium oxide is an alkali metal oxide with the chemical formula Na2O. It is the anhydride form of sodium hydroxide. When sodium burns in the air it burns with a bright, white light.
What is the nature of sodium oxide?
Sodium oxide is a simple strongly basic oxide. It is basic because it contains the oxide ion, O2-, which is a very strong base with a high tendency to combine with hydrogen ions. Reaction with water: Sodium oxide reacts exothermically with cold water to produce sodium hydroxide solution.
What happens if you breathe in lye fumes?
Breathing in sodium hydroxide causes irritation of eyes, nose and throat, cough, chest tightness, headache, fever and confusion. In serious cases damage to the airways, a fast Page 3 Compendium of Chemical Hazards: Sodium Hydroxide Page 3 of 4 General Information heart rate and eye damage can occur.
Is sodium oxide a base or alkali?
A base is a substance that can react with acids and neutralise them. Bases are usually: metal hydroxides, such as sodium hydroxide, or….Bases v alkalis.
| Copper oxide | Sodium hydroxide | |
|---|---|---|
| Is it an alkali? | No | Yes |
What is the electronegativity difference of sodium oxide?
The difference in electronegativity between sodium(0.93) and oxygen(3.44) is 3.44–0.93=2.51 which is higher than 1.7, hence, according to the Pauling scale, the bond formed between sodium and an oxygen atom is ionic in nature.
Why is sodium oxide Na2O and not NaO?
NaO is a formula, so that must be the formula of NaO. Na2O is more basic because by reacting with water it forms strong base, NaOH whereas MgO will form Mg(OH)2 which is a weaker base. Oxides are divided into 2 categories – metallic and non metallic. Oxides are divided into 2 categories – metallic and non metallic.
What happens if you touch sodium?
It can hurt you if it touches your skin, if you drink it or if you breathe it. Eating or drinking sodium hydroxide can cause severe burns and immediate vomiting, nausea, diarrhea or chest and stomach pain, as well as swallowing difficulties.
What are the properties of sodium oxide?
Properties of Sodium oxide Na2O: White, heat-resistant, refractory. Shows strong basic properties, reacts vigorously with water (forms an alkaline solution), acids, acidic and amphoteric oxides, liquid ammonia. Obtaining sodium oxide: 2 Na + 2 NaOH = 2 Na2O + H2 (600°C).
How does sodium oxide react with water?
Sodium oxide is a simple strongly basic oxide. It is basic because it contains the oxide ion, O2-, which is a very strong base with a high tendency to combine with hydrogen ions. Reaction with water: Sodium oxide reacts exothermically with cold water to produce sodium hydroxide solution.
What is the Lewis structure for sodium oxide?
The formation of Na2O can be shown with the help of Lewis dot structure. Na+ is the sodium cation and O2- is the oxide anion. Na has one electron in its valence shell, which it loses to achieve a noble gas configuration while oxygen has 6 electrons in its valence shell, 2 short of achieving a noble gas configuration.
What is the chemical formula for sodium oxide?
Sodium oxide is an alkali metal oxide with the chemical formula Na 2 O. It is the anhydride form of sodium hydroxide. When sodium burns in the air it burns with a bright, white light. When it burns in oxygen it burns even more brightly. It forms a white powder called sodium oxide.



