What are the rules for naming ethers?
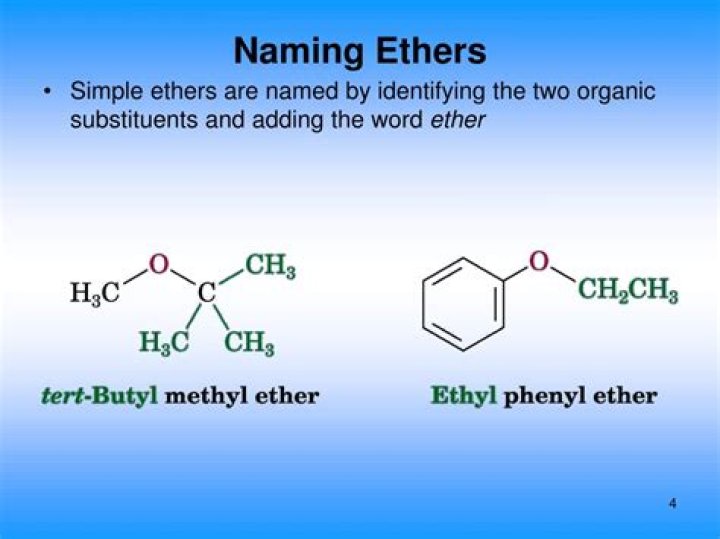
Common names of ethers simply give the names of the two alkyl groups bonded to oxygen and add the word ether. The current practice is to list the alkyl groups in alphabetical order (t-butyl methyl ether), but older names often list the alkyl groups in increasing order of size (methyl t-butyl ether).
Do ethers get priority in naming?
If a substituent is an alcohol, the alcohol has higher priority. However, if a substituent is a halide, ether has higher priority. If there is both an alcohol group and a halide, alcohol has higher priority. The numbering begins with the end that is closest to the higher priority substituent.
How do you name compounds with substituents?
In summary, the name of the compound is written out with the substituents in alphabetical order followed by the base name (derived from the number of carbons in the parent chain). Commas are used between numbers and dashes are used between letters and numbers. There are no spaces in the name.
How do you name ethers with a benzene ring?
IUPAC nomenclature names ethers as alkoxy alkanes, alkoxy alkenes, or alkoxy alkynes. The group in the chain that has the greatest number of carbon atoms is designated the parent compound. In the case of aromatic ethers, the benzene ring is the parent compound.
What is an ether substituent?
Ethers are compounds having two alkyl or aryl groups bonded to an oxygen atom, as in the formula R1–O–R2. The ether functional group does not have a characteristic IUPAC nomenclature suffix, so it is necessary to designate it as a substituent.
Is ether a substituent or functional group?
The key difference between functional group and substituent is that a functional group is an active part of a molecule whereas a substituent is a chemical species that can replace an atom or a group of atoms in a molecule. In fact, they are a specific type of substituents.
How do you name ethers and esters?
The ethers are named as alkyl alkyl ethers, with the alkyl groups in alphabetical order followed by the class name ether, each as a separate word. Thus, CH3OCH2CH2CH3 is methyl propyl ether. Esters are named as alkyl alkanoates.
How do you name benzene?
BenzeneBenzene / IUPAC ID
What are substituents in organic chemistry?
Substituent: An atom or group other than hydrogen on a molecule. (The atom or group has substituted for the missing hydrogen.) Cyclohexane has no substituents.
How do you name amines with substituents?
More complex primary amines are named with —NH2 as the amino substituent. Aromatic amines: named as derivatives of the parent compound aniline. Substituents attached to the nitrogen are indicated by using “N-” as the location number.
What is the nomenclature of substituted benzene?
Substituted Benzene Compounds: Nomenclature. Benzene is a hydrocarbon with the chemical formula C 6 H 6. It has 6 carbon atoms joined in a ring and has 1 hydrogen atom attached to each of the carbon atoms. By replacing one or more of the hydrogen atoms with some functional group, we get several benzene compounds.
How do you name a compound with a benzene ring?
Similar to the base name nomenclature system, the carbon in which the base substituent is attached on the benzene ring is given the first priority and the rest of the substituents are given the lowest number order possible. Under this consideration, the above compound can be named: 2,4-difluorobenzyl chloride.
What is the nomenclature of ethers?
Nomenclature of Ethers. Ethers are compounds having two alkyl or aryl groups bonded to an oxygen atom, as in the formula R 1–O–R 2.
How do you name a compound with a benzyl group?
2- (methylene) group attached to the benzene ring. This group can be used to name particular compounds, such as the one shown below. This compound has chlorine attached to a benzyl group, therefore it is called benzyl chloride. Benzoyl = Bz = .



