What are the shape of hybrid orbitals?
The five basic shapes of hybridization are linear, trigonal planar, tetrahedral, trigonal bipyramidal, and octahedral. The geometry of the orbital arrangement: Linear: Two electron groups are involved resulting in sp hybridization, the angle between the orbitals is 180°.
What is the shape of SP sp2 Sp3 and sp3d hybrid orbitals?
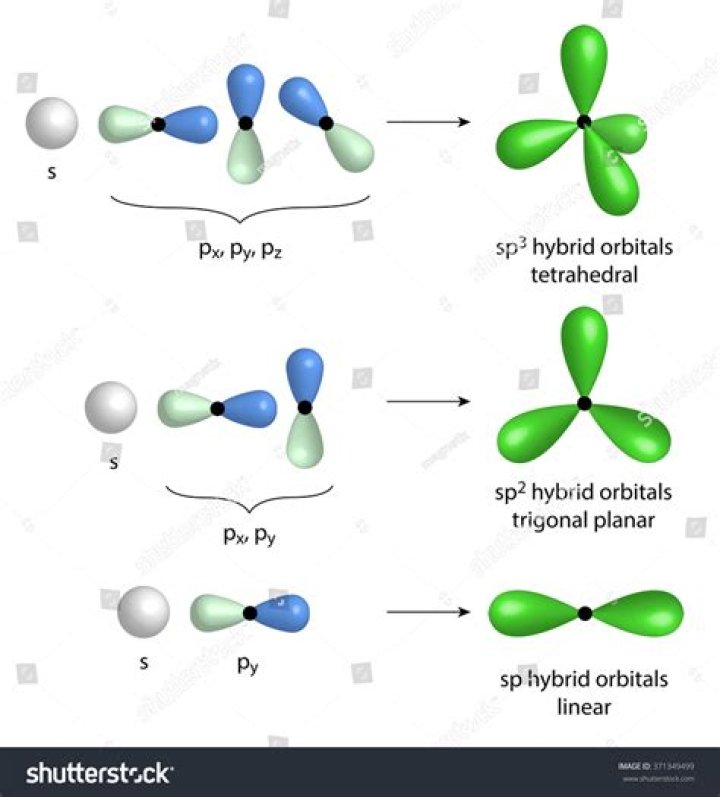
It has linear shape. Bond angle is 180°. sp2 hybridisation: One s-and two p-orbitals get hybridised to form three equivalent hybrid orbitals. The three hybrid orbitals are directed towards three corners of an equilateral triangle.
What are the 3 hybrid orbitals?
| Number of Groups around the atom | Sub-Shapes A = central atom X = atom attached to central atom E = nonbonding electron group on central atom | Hybridization |
|---|---|---|
| 3 | AX3, Trigonal Planar AX2E, Bent | sp2 |
| 4 | AX4, Tetrahedral AX3E, Trigonal Pyramidal AX2E2, Bent | sp3 |
| 5 | AX5, Trigonal Bipyramidal AX4E, See-Saw AX3E2, T-Shaped AX2E3Linear | sp3d |
What is the shape of sp2 hybrid?
For sp2 hybridized central atoms the only possible molecular geometry is trigonal planar. If all the bonds are in place the shape is also trigonal planar. If there are only two bonds and one lone pair of electrons holding the place where a bond would be then the shape becomes bent.
What is the shape of sp3d hybridization?
trigonal bipyramid
The geometrical shape of sp3d hybridisation is trigonal bipyramid.
How do you identify hybrid orbitals?
The carbon is bonded to two other atoms, that means it needs two hybrid orbitals, aka sp. An easy way to figure out what hybridization an atom has is to just count the number of atoms bonded to it and the number of lone pairs. Double and triple bonds still count as being only bonded to one atom.
What are the examples of hybridization?
EXAMPLES – TYPES – HYBRIDIZATION IN CHEMISTRY
- sp hybridization examples (Beryllium chloride, BeCl2; Acetylene, C2H2)
- sp2 (Boron trichoride, BCl3; Ethylene, C2H4)
- sp3 (Methane, CH4; Ethane, C2H6)
- sp3d (phosphorus pentachloride, PCl5)
- sp3d2 (sulfur hexafluoride, SF6)
- sp3d3 (Iodine heptafluoride, IF7)
What is the shape of Sp3 hybrid orbital?
sp 3 hybrid orbitals are oriented at bond angle of 109.5 o from each other. This 109.5 o arrangement gives tetrahedral geometry (Figure 4). Because carbon plays such a significant role in organic chemistry, we will be using it as an example here.
What are the shapes and geometry of Sp2 and Sp3 hybrid orbital’s?
i Sp-hybrid orbital is oriented to an angle 180° . ii Sp2 -hybrid orbital lie in a plane and is directed towards the corners of equilateral triangle making an angle of 120°iii Sp3 -hybrid orbitals are directed towards the four corners of tetrahedron making an angle of 109° 28.
What are examples of hybrid orbitals?
Carbon is a perfect example showing the need for hybrid orbitals. As you know, Carbon’s ground state configuration is: According to Valence Bond Theory , carbon should form two covalent bonds, resulting in a CH2, because it has two unpaired electrons in its electronic configuration.
What are the shapes of orbitals?
The angular quantum number (l) describes the shape of the orbital. Orbitals have shapes that are best described as spherical (l = 0), polar (l = 1), or cloverleaf (l = 2). They can even take on more complex shapes as the value of the angular quantum number becomes larger.
What is an example of orbital hybridization?
Explanation: Methane is an example of sp3 hybridization. When one s and 3 p orbitals get mixed or hybridized and form 4 sp3 hybridized orbitals, it is called sp3 hybridization. Carbon atom has 1 s and 3 p (Px, Py, Pz) orbitals in its outermost shell.So these 4 orbitals hybridize and form 4 sp3 hybridized orbitals.
What are sp hybridized orbitals?
In sp hybridization, the s orbital overlaps with only one p orbital . Atoms that exhibit sp hybridization have sp orbitals that are linearly oriented; two sp orbitals will be at 180 degrees to each other. Any central atom surrounded by just two regions of valence electron density in a molecule will exhibit sp hybridization.



