What are the three bond angles in the trigonal bipyramidal structure?
What are the bond angles of a molecule with trigonal bipyramidal geometry? When a molecule has trigonal bipyramidal geometry bond angles are 120°, 90°.
What are the ideal bond angles for a trigonal bipyramidal molecule?
| Trigonal bipyramidal molecular geometry | |
|---|---|
| Point group | D3h |
| Coordination number | 5 |
| Bond angle(s) | 90°, 120° |
| μ (Polarity) | 0 |
What is the molecular geometry of CH3Cl?
tetrahedral
Thus, CH3Cl’s molecular geometry is tetrahedral, and its bond angles are 109.5°.
What is bond angle?
A bond angle is the angle between two bonds originating from the same atom in a covalent species. eg. Geometrically, a bond angle is an angle between two converging lines.
What is smallest bond angle?
H2O has smallest bond angle. Due to repulsion between two lone pairs bonds are displaced slightly which results in decrease in bond angle.
What is the bond angle of CH3Cl?
110.5°
The (HCH) bond angle in CH3Cl is 110.5°. The HCH bond angle is wider than in CH4 which suggests that the C–Cl single bond takes up less space close to the carbon. This is due to the C–Cl bonds polarity with δ– on the chlorine, chlorine being more electronegative than carbon (and hydrogen).
What bonds are in CH3Cl?
This is an ionic compound because the chlorine atom is attached to the carbon atom, so the electronegativity difference is more. So there is an ionic bond that exists between C-Cl.
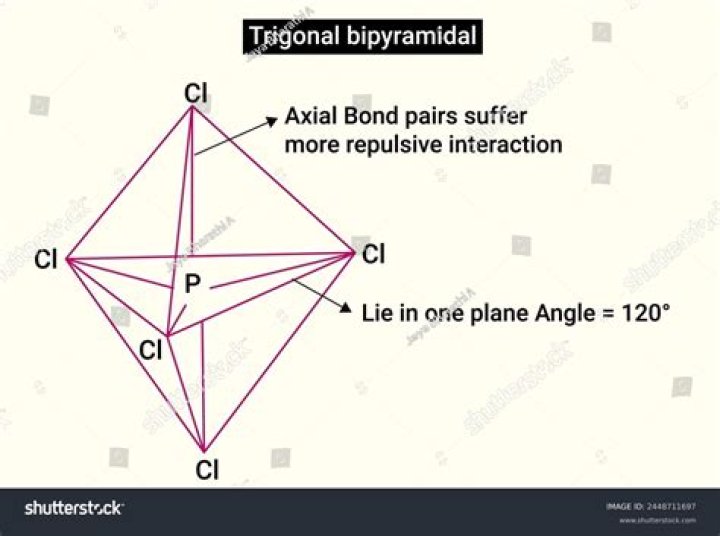
What is the bond angle of trigonal bipyramidal?
The Trigonal Bipyramidal is a molecular shape where there are 5 bonds attached to a central atom. There are two bond angles for this shape. The first one is 90 degrees and the second one is 120 degrees.
What are the stereoisomers of trigonal bipyramidal structure?
Since there are two types of atoms on a Trigonal Bipyramidal structure, axial and equatorial, there are different Stereoisomers that could arise depending on the substituents attached. For example, if there is 4 X atoms and 1 Y atom attached to the central atom, Y could either be in an equatorial position or in an axial position.
What is trigonal bipyramidgeometry?
The Chlorine atoms are as far apart as possible at nearly 90oand 120obond angle. This is trigonal bipyramidgeometry. Trigonal bipyramid geometry is characterized by 5 electron pairs. Sulfur Tetrafluoride: Chime in new window
What are the bond angles of a see saw molecule?
Since there are only 5 bonds attached to it and no lone pairs, the bond angles tend to be 90 degrees and 120 degrees since the bonds won’t bend. The See-Saw is a molecular shape where there are 4 bonds attached to the central atom along with 1 lone pair.



