What causes mass independent fractionation?
Most isotopic fractionations (including typical kinetic fractionations and equilibrium fractionations) are caused by the effects of the mass of an isotope on atomic or molecular velocities, diffusivities or bond strengths. …
What is mass dependent isotope fractionation?
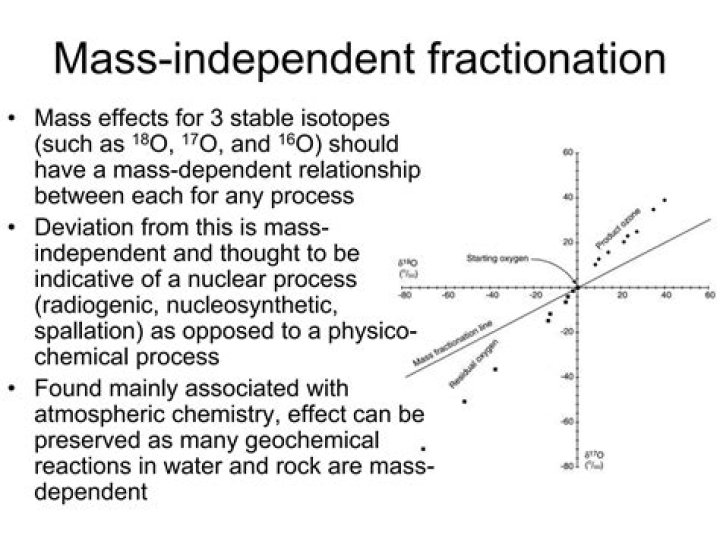
Isotopic variations usually follow mass-dependent fractionation, meaning that the relative variations in isotopic ratios scale with the difference in mass of the isotopes involved (e.g., δ17O ≈ 0.5 × δ18O).
Why does isotopic fractionation occur?
Isotopic fractionations are related to bond strength. Thus, stronger bonds generally favor heavier isotopes. A difference in the bond strengths of complexes in a ligand exchange reaction thus results in isotopic fractionation.
What are the masses of the stable isotopes of sulfur?
Sulfur (16S) has 23 known isotopes with mass numbers ranging from 27 to 49, four of which are stable: 32S (95.02%), 33S (0.75%), 34S (4.21%), and 36S (0.02%)….List of isotopes.
| n: | Neutron emission |
|---|---|
| p: | Proton emission |
What is fractionation factor?
The fractionation factor is the factor by which the abundance ratio of two isotopes will change during a chemical reaction or a physical process.
What is the terrestrial fractionation line?
d18O for a given body gives a linear array of data points which is characteristic of the bulk composition of the body. For the Earth the array is termed the Terrestrial Fractionation Line (TFL). Thus, the slope and intercept of a planetary body’s oxygen three-isotope fractionation line are definitive.
What is equilibrium isotope fractionation?
Equilibrium isotope fractionation is the partial separation of isotopes between two or more substances in chemical equilibrium. Isotopic fractionations attributed to equilibrium processes have been observed in many elements, from hydrogen (D/H) to uranium (238U/235U).
How do you find fractionation?
First, multiply the whole number by the denominator of the fractional part. Take the resulting figure and add it to the fraction’s numerator. Take this final figure as your new numerator and place it over the original denominator. This gives you your improper fraction.
What is the purpose of isotopic enrichment?
A process by which the relative abundance of the isotopes of a given element are altered, thus producing a form of the element that has been enriched in one particular isotope and depleted in its other isotopic forms.
What is isotopic equilibrium?
At isotopic equilibrium, the forward and backward reaction rates of any particular isotope are identical. This does not mean that the isotopic compositions of two compounds at equilibrium are identical, but only that the ratios of the different isotopes in each compound are constant for a particular temperature.
What is the mass of the most common isotope of sulfur?
sulfur-32
| ChEBI Name | sulfur-32 atom |
|---|---|
| Definition | The stable isotope of sulfur with relative atomic mass 31.972071. The most abundant (95.02 atom percent) isotope of naturally occurring sulfur. |
| Stars | This entity has been manually annotated by the ChEBI Team. |
| Supplier Information | |
| Download | Molfile XML SDF |
How many unstable isotopes does sulfur have?
Radioactive sulfur isotopes. Sulfur has 14 unstable isotopes. Radioactive sulfur with mass 35 is formed in the atmosphere from cosmic ray spallation of argon with mass 40 and has a half-life of 87 days. Therefore, it is used in hydrological studies of biologically less active surface environments.
Is there mass-independent fractionation of sulfur?
Mass-independent fractionation of sulfur can be observed in ancient sediments, where it preserves a signal of the prevailing environmental conditions.
What is mass-independent isotope fractionation?
Mass-independent isotope fractionation or Non-mass-dependent fractionation (NMD), refers to any chemical or physical process that acts to separate isotopes, where the amount of separation does not scale in proportion with the difference in the masses of the isotopes.
What is mass-independent fractionation in Ozone?
Mass-independent fractionation also has been observed in ozone. Large, 1:1 enrichments of 18 O/ 16 O and 17 O/ 16 O in ozone were discovered in laboratory synthesis experiments by John Heidenreich and Mark Thiemens in 1983, and later found in stratospheric air samples measured by Konrad Mauersberger.
What happened to non-mass-dependent NMD fractionations in sedimentary rocks?
The disappearance of distinctive non-mass-dependent (NMD) sulphur isotope fractionations in sedimentary rocks deposited after about 2.4–2.3 Gyr ago16 (Fig. 2). Almost all fractionations among isotopes of a given element scale to differences in their masses; NMD fractionations deviate from this typical behaviour.



