What causes the Colour observed in para red?
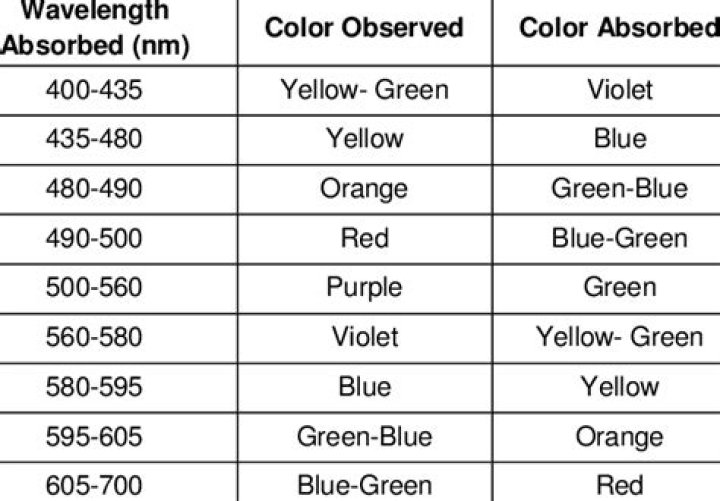
Color changes are caused by change in extent of delocalization of electrons. More delocalization shifts the absorption max to longer wave lengths and makes the light absorbed redder, while less delocalization shifts the absorption max to shorter wavelengths.
What is para red used for?
Para Red is a biological stain. It is used as a fat soluble chemical dye and food additive. It is a banned colorant and a number of analytical techniques have been developed to detect it in food.
Which of the following reagents are required for preparation of P red from nitroaniline?
Draw the mechanism for the synthesis of para red. Reagents: 4-nitroaniline, conc. H2SO4, sodium nitrite, and water.
What is Para Red dye?
Para Red (paranitraniline red, Pigment Red 1, C.I. 12070) is a dye. It was discovered in 1880 by von Gallois and Ullrich and was the first azo dye. It dyes cellulose fabrics a brilliant red color, but is not very fast. The dye can be washed away easily from cellulose fabrics if not dyed correctly.
What are the components of dye?
In addition to chromophores, most dyes also contain groups known as auxochromes (colour helpers), examples of which are carboxylic acid, sulfonic acid, amino, and hydroxyl groups.
Which chemical is used in the manufacture of dyes?
The basic raw materials used for the manufacture of dyestuff are Benzene, Toluene, Xylene and Naphthalene (BTXN). These raw materials are initially transformed into dye intermediates by nitration, sulphonation, amination, reduction and other chemical unit process.
What is the Iupac name of Para Red?
| Para Red | |
|---|---|
| IUPAC name | 1-[(E)-(4-Nitrophenyl)diazenyl]-2-naphthol |
| Other names | 1-((4-Nitrophenyl)azo)-2-naphthalenol, 1-((4-nitrophenyl)azo)-2-naphthol, 1-((p-nitrophenyl)azo)-2-naphthalenol, 1-((p-nitrophenyl)azo)-2-naphthol, paranitraniline red, Pigment Red 1, C.I. 12070, Recolite Para Red B, Carnelio Para Red BS |
| Identifiers |
How can we prepare para nitroaniline from aniline?
Dissolve 4.0 mL of aniline in 10 mL of acetic acid in a 100 mL round bottom flask. To this solution, add 5 mL of acetic anhydride and mix well by swirling. CAUTION: the reaction is exothermic and the flask becomes warm.
How do you get para nitroaniline aniline?
p-nitroaniline : Acetanilide on treatment with nitric acid in the presence of sulphuric acid gives p-nitroacetanilide which on acidic hydrolysis gives p-nitroaniline as major product.
How dyes are manufactured?
Dyes are synthesized in a reactor, filtered, dried, and blended with other additives to produce the final product. In general, organic compounds such as naphthalene are reacted with an acid or an alkali along with an intermediate (such as a nitrating or a sulfonating compound) and a solvent to form a dye mixture.
How do you make para red solution?
Add the cloth to 50 mL of a alkaline 2-naphthol solution. After several minutes, remove the cloth and rinse it repeatedly with water. Synthesis of Para Red: Dissolve 2.7 g of 2-naphthol in 50 ml of 2.5M NaOH solution, Cool the solution to 10 °C by adding crushed ice.
How do you make Para Red from nitroaniline?
Synthesis of Para Red, part 1 In this lab, you prepare p-nitroaniline by the nitration of acetanilide and subsequent hydrolysis of the nitration product. You will then diazotize this compound and use the diazonium salt to synthesize the dye Para Red.
How do you make Para Red from naphthol?
Synthesis of Para Red: Dissolve 2.7 g of 2-naphthol in 50 ml of 2.5M NaOH solution, Cool the solution to 10 °C by adding crushed ice. Carefully pour this solution into the flask containing the remainder of the diazonium salt, while swirling. Continue to swirl the mixture vigorously for a few minutes, then acidify it with 1 M sulfuric acid.
How do you make para red dye with NaOH?
Dyeing Cloth Samples with Para Red: Place 0.5 g of 2-naphthol in a beaker containing 100 mL of hot water. Add 2.5 M NaOH solution dropwise while swirling the mixture until about three-fourths of the 2-naphthol dissolves.



