What does a Tafel plot tell you?
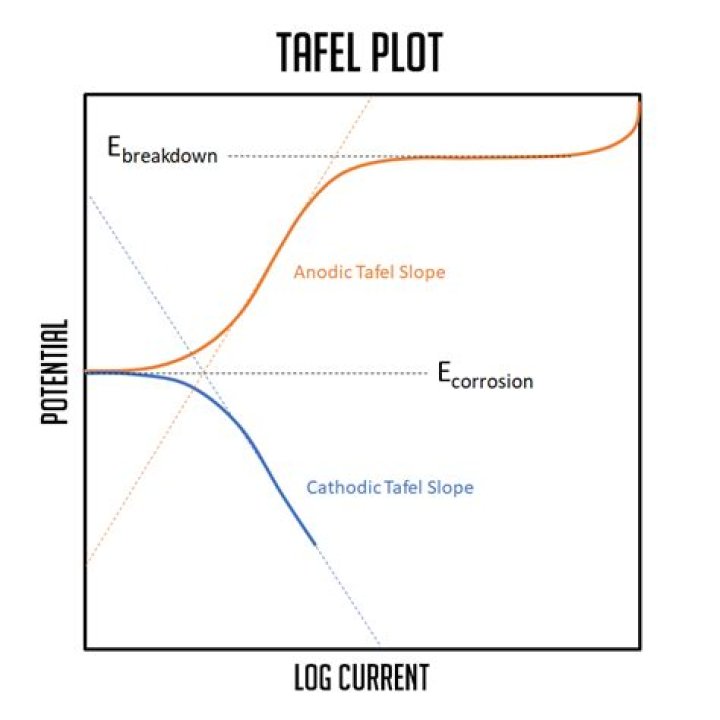
A Tafel plot is a graphical plot (usually logarithmic) showing the relationship between the current generated in an electrochemical cell and the electrode potential of a specific metal. These plots are usually generated based on electrochemical experiments performed under controlled conditions.
How is Tafel plot calculated?
take log value of abs current density data 4. plot the log value as x axis and potential as y axis. Zoom the graph in the kinetically controlled region. You will get the tafel plot.
What is Tafel slope unit?
mV/dec is the Tafel slope unit in electrochemistry. It basically evaluates how much overpotential we need to increase the rate of reaction by ten times on the respective electrode. Generally, Pt has 120mV/dec to 125mV/dec. It means you have to increase the potential by 120mV for per decad of current density.
What is the Tafel region?
Remember a Tafel region is a region of the E-log i curve that is straight over a significant range. If you are comparing corrosion rate from tafel extrapolation and corrosion rate measured by weight loss, then it is expected to have an error .
What is Tafel law?
The Tafel equation is an equation in electrochemical kinetics relating the rate of an electrochemical reaction to the overpotential. The Tafel equation was first deduced experimentally and was later shown to have a theoretical justification.
What is Tafel behavior?
The Tafel equation describes the behavior of one isolated reaction. In a corrosion system, we have two opposing reactions: anodic and cathodic. The Tafel equations for the anodic and cathodic reactions in a corrosion system can be combined to generate the Butler-Volmer equation (Eq. 2).
What are Tafel constants?
It is proposed that Tafel constant can be considered as the onset potential of HER. Tafel constant becomes the defining parameter between two electrocatalysts when other parameters such as Tafel slope or exchange current density become same.
What is the use of Tafel slope?
The tafel slope shows how efficiently an electrode can produce current in response to change in applied potential. So if the slope (mV/decade) is lower means less overpotential is required to get high current.
What theory did Tafel propose?
The Tafel equation is an equation in electrochemical kinetics relating the rate of an electrochemical reaction to the overpotential. The Tafel equation was first deduced experimentally and was later shown to have a theoretical justification. The equation is named after Swiss chemist Julius Tafel.
What is the Stern Geary equation?
The Stern-Geary Equation describes the relationship between the Polarization Resistance (Rp) and the Corrosion Current (iCORR). IMPORTANT: The Stern-Geary Equation is derived from the Butler-Volmer Equation via a series expansion in which ΔE /βa,c is assumed to be less than 0.1.
How do you plot Tafel plot?
Popular Answers (1)
- take one LSV curve at a particular rotation rate.
- convert the current to current density and the take absolute value of the current density data.
- take log value of abs current density data.
- plot the log value as x axis and potential as y axis. Zoom the graph in the kinetically controlled region.
What is the temperature of a Tafel plot?
Tafel plot, where and are both chosen to be 0.5 the temperature is 298.15 K. A iadicates the linear region (eq. 23) and B the Tafel (eq. 24). FIGURE 1-8 Tafel plots for the cathodic and anodic branches of the current-potential curve.
What is a Tafel plot in electrochemistry?
A Tafel plot is a graphical plot (usually logarithmic) showing the relationship between the current generated in an electrochemical cell and the electrode potential of a specific metal.
How do you find the value of a in Tafel equation?
The values of constants a and b in the Tafel equation can be calculated by comparing Eqs. (8.6) and (8.7) with Eq. (8.10). A graph, known as a Tafel plot, can be drawn representing the relationship between the overpotential and the logarithmic current density.
What is the Tafel extrapolation used for?
Tafel extrapolation of polarization curves is used to determine the corrosion current density, icorr (mA cm − 2) from which it is possible to determine the corrosion rate, Pi (mm year − 1) from 76: (10) P i = 11.85 i corr



