What does sodium sulfite react with?
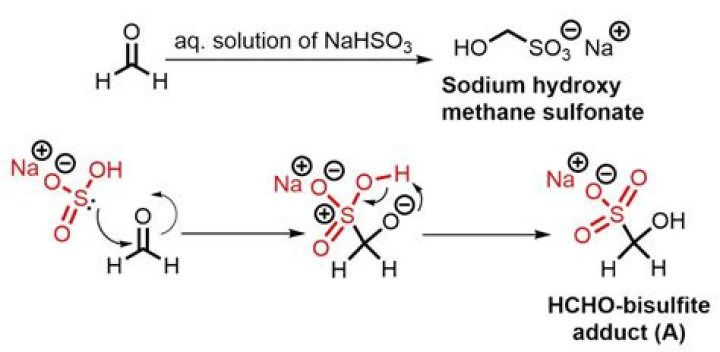
Sodium sulfite reacts with aldehydes to yield a bisulfite adduct. However, sulfonic acids are produced from its reaction with ketones. Solutions of sodium sulfite are oxidized by atmospheric oxygen, yielding sodium sulfate. This compound is insoluble in ammonia and chlorine.
Does sodium sulfite remove chlorine?
As a result, dechlorination was instituted to remove residual chlorine from wastewater prior to discharge into sensitive aquatic waters. Typically, dechlorination is accomplished by adding sulfur dioxide or sulfite salts (i.e., sodium sulfite, sodium bisulfite, or sodium metabisulfite).
Does sodium bisulfate neutralize bleach?
Sodium Metabisulfite Water treatment plants employ the substance to remove trace of excessive chlorine. Sodium metabisulfite is an effective bleach neutralizer. 2.2 grams (one teaspoon) of sodium metabisulfite added to 2.5 gallons of water effectively neutralizes all harmful bleach residue.
What Cannot be mixed with sodium hypochlorite?
Sodium hypochlorite reacts with ammonia, drain cleaners, and other acids. Many household products state that they contain bleach on the label. Pool chemicals frequently containing calcium hypochlorite or sodium hypochlorite, should not be mixed with household cleaners, and used carefully.
Is sodium sulfite toxic?
Sodium sulfite is a white odorless powder. Density 2.633 g / cm3. Moderately toxic.
Is sodium sulfite corrosive?
Soluble in water. Reacts with hot water, steam or acids to produce corrosive material.
What happens when you mix sodium hypochlorite and sodium bisulfite?
Chemical interactions Mixing anything with hypochlorite can be extremely dangerous. Contact between hypochlorite and other treatment plant chemicals such as alum, ferric, or sodium bisulfate, can cause an uncontrolled explosive release of energy, heat, and chlorine gas. A mistake in chemical delivery can be disastrous.
How do you neutralize sodium chlorite?
If a specific situation requires sodium chlorite to be neutralized, the chlorite must first be reduced by a reaction with sodium sulfite. The use of sodium sulfite is recommended over other reducing agents such as sodium thiosulfate (Na2S2O3), sodium bisulfite (NaHSO3), and sodium meta-bisulfite (Na2S2O5).
How do I make a bleach neutralizer?
The neutralizing solution is made by mixing 1 part hydrogen peroxide to 10 parts water. Mix this up first before you begin your bleaching project, so it is ready to use. The solution can be mixed in a plastic tub, the kitchen sink, or in your washer.
What happens when you mix salt and bleach?
When dissolved in water, bleach provides OH-. There will be a neutralization reaction if the salt you use is acidic. There could be a reaction if the salt you use is basic. The salt will be dissolved in a bleach solution.
What happens when you mix sodium sulfite and bleach?
Beside above, what happens when you mix sodium sulfite and bleach? The exothermic reaction of sodium hypochlorite (bleach) with sodium sulfite produces heat. The negative sign is necessary because if heat is absorbed by solution then ∆Treaction is positive and then the qreaction must be exothermic and negative.
What is the reaction between sodium hypochlorite and sodium sulfite?
The exothermic reaction of sodium hypochlorite (bleach) with sodium sulfite produces heat. The negative sign is necessary because if heat is absorbed by solution then ∆Treaction is positive and then the qreaction must be exothermic and negative.
What is sulfur dioxide used for in Bleach?
Sulfur dioxide and its derivatives have been used to bleach textiles since earliest times. Besides being an important bleaching agent in the pulp and paper industry, sulfur dioxide is also used in the manufacture of chlorine dioxide, sodium hydrosulfite, and sodium sulfite. When SO2 is dissolved in water]
What is the function of sulfur in sodium thiosulfate?
This compound is also an important component in the preparation of sodium thiosulfate. Sulfites are compounds containing the sulfite ion, most often in combination with sodium (sodium sulfite) or potassium (potassium sulfite). Sulfites release the irritant gas sulfur dioxide, which acts as a preservative and bleaching agent.



