What happens to iron in a redox reaction?
The following is an oxidation/reduction reaction (redox) reaction. Iron is Fe and Fe2O3 is red rust. It has gone from its elemental state with no charge ( Fe0) to its ionic state (Fe3+) Because the iron has lost electrons and become positively charged, it has been oxidized. The oxygen has been reduced.
When iron is oxidized What is reduced?
The oxidation state of iron goes from {0 3+}; iron is therefore the reducing agent. Note: knowing that oxygen, a common oxidizing agent, is typically reduced to {2–} allows us to deduce the oxidation state of iron in the product to be {3+} (from Fe2O3).
What is reduction in redox reaction?
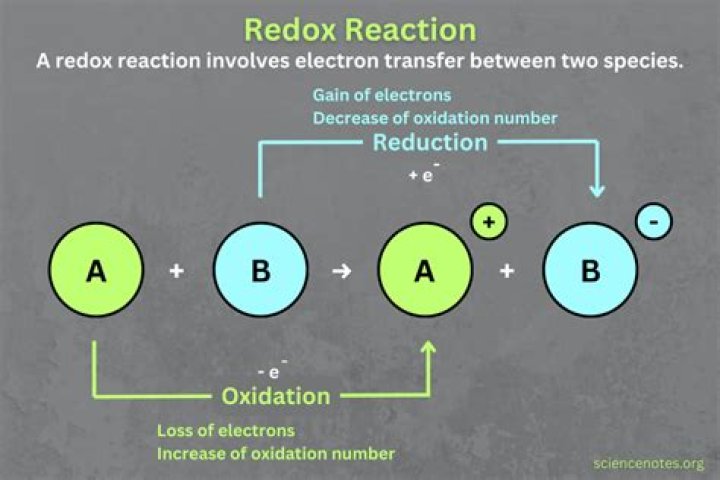
An oxidation-reduction (redox) reaction is a type of chemical reaction that involves a transfer of electrons between two species. An oxidation-reduction reaction is any chemical reaction in which the oxidation number of a molecule, atom, or ion changes by gaining or losing an electron.
Is rusting of iron a redox reaction?
Rusting of iron is an example of a redox reaction. This is an oxidation reaction where oxygen acts as an oxidising agent. Since oxygen also combines with the metal iron, this is a reduction reaction, where the metal iron acts as a reducing agent.
What happens when ferrous sulphate is added to potassium permanganate?
When potassium permanganate is added to the acidulated solution of ferrous sulphate, ferrous ion is oxidised and the acid is neutralised.
How does redox reaction occur?
An oxidation reaction strips an electron from an atom in a compound, and the addition of this electron to another compound is a reduction reaction. Because oxidation and reduction usually occur together, these pairs of reactions are called oxidation reduction reactions, or redox reactions.
What is a redox reaction example?
The formation of hydrogen fluoride is an example of a redox reaction. Another example of a redox reaction is the formation of hydrogen fluoride. The hydrogen is oxidized and loses two electrons, so each hydrogen becomes positive. The two electrons are gained by fluorine, which is reduced.
What is the overall reaction between permanganate and MnO4?
The overall reaction is: 8H+ (aq) + MnO4- (aq) + 5e- Mn2+ (aq) + 4H2O In acid solutions the permanganate ion undergoes reduction to manganese (2+) ion.
What are the reactants and products of permanganate and iron oxide?
In the example given, the two reactants are permanganate (MnO4-) and iron (Fe2+), and the products are manganese (Mn2+) and iron (Fe3+). It is given that the reaction is occurring under acidic conditions, so we add H+ ions to the left side and water on the right side to balance the oxygen atoms that are present.
How do oxalic and sulfuric acid react with permanganate?
At first, when the permanganate is added to the titration flask containing both oxalic and sulfuric acid the solution The sulfuric acid added to the titration flask acts as a proton donor for the solution permanganate ion acts as an indicator itself. The reaction produces its own catalyst in the form of Mn 2+ that promotes oxidation.
How to titrate a reduced sample with permanganate solution?
Add reduced sample to the flask. Titrate with permanganate solution until a faint pink color persists for 30 seconds. According to the reaction equation given above 1 mole of permanganate reacts with 5 moles of iron and this ratio have to be used for titration result calculation.



