What has the highest boiling point propane butane methane or butane?
The Saturated Hydrocarbons, or Alkanes
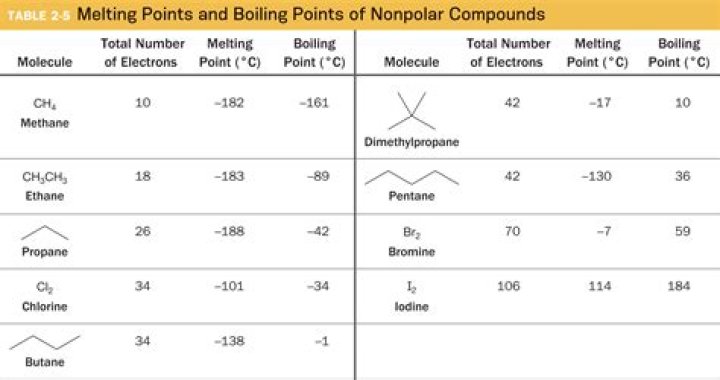
| Name | Molecular Formula | Boiling Point (oC) |
|---|---|---|
| methane | CH4 | -164 |
| ethane | C2H6 | -89 |
| propane | C3H8 | -42 |
| butane | C4H10 | -0.5 |
Which alkane has the highest boiling point in order?
1. Nonane will have a higher boiling point than octane, because it has a longer carbon chain than octane. 2. Octane will have a higher boiling point than 2,2,3,3‑tetramethylbutane, because it branches less than 2,2,3,3‑tetramethylbutane, and therefore has a larger “surface area” and more van der Waals forces.
What are the boiling points of methane and hexane?
A totally symmetrical molecule like methane is completely non-polar, meaning that the only attractions between one molecule and its neighbors will be Van der Waals dispersion forces….Boiling Points.
| Molecular Name | hexane |
|---|---|
| Formula | C 6H 14 |
| Melting Point (°C) | –95 |
| Boiling Point (°C) | 69 |
| Density (20°C)* | 0.659 g/mL |
Which has higher boiling point hexane or pentane?
In the case of hexane, the carbon atom chain is of 6 atoms while in the case of pentane it is of 5 atoms. Hence, the surface area of hexane is larger than the pentane. So, it will have a higher boiling point than pentane.
Does propane have a higher boiling point than hexane?
Increasing the contract surface area between hydrocarbons raises boiling point, so hexane should have a higher boiling point than propane.
Which has a higher boiling point ethane or butane?
Stronger intermolecular forces take more energy to overcome therefore the stronger the intermolecular forces, the higher the boiling point. This means that butane will have a higher boiling point than ethane.
Which has the highest boiling point methane ethane or propane?
Because propane is larger there will be more London attraction forces holding propane molecules together compared to ethane and methane, so propane will have higher melting and boiling points.
Does butane have a high boiling point?
30.2°F (-1°C)
Butane/Boiling point
What is the boiling point of butane?
Does pentane have a higher boiling point than propane?
These forces are generally stronger with increasing molecular mass, so propane should have the lowest boiling point and n-pentane should have the highest, with the two butane isomers falling in between.
What is boiling point of ethane?
-128.2°F (-89°C)
Ethane/Boiling point
Why does pentane have a higher boiling point than butane?
Alkanes are covalent molecules with weak van der Waals forces between. The larger the molecule, the more van der Waals forces there are and the higher the boiling point. So pentane is higher than butane. In some courses London forces are mentioned.
What is the boiling point of a straight chain alkane?
As a rule of thumb, the boiling point rises 20 – 30 °C for each carbon added to the chain; this rule applies to other homologous series. A straight-chain alkane will have a boiling point higher than a branched-chain alkane due to the greater surface area in contact, thus the greater van der Waals forces, between adjacent molecules.
What is the name of the molecular formula for butane?
Name Molecular Formula Melting Point (oC) Boiling Point (oC) butane C4H10 -138 -0.5 pentane C5H12 -130 36 hexane C6H14 -95 69 heptane C7H16 -91 98
What is the boiling point of isobutane?
For example, compare isobutane (2-methylpropane) and n-butane (butane), which boil at -12 and 0 °C, and 2,2-dimethylbutane and 2,3-dimethylbutane which boil at 50 and 58 °C, respectively.



