What ions does ammonium produce?
Anhydrous, or gaseous, NH3 is a very important direct-application N fertilizer. Gaseous NH3, when in contact with moist soil, dissolves in, and reacts with, soil water to form NH4+ and OH-ions.
What is NH4+ in chemistry?
The ammonium cation is a positively charged polyatomic ion with the chemical formula NH + 4. It is formed by the protonation of ammonia (NH. 3). Ammonium is also a general name for positively charged or protonated substituted amines and quaternary ammonium cations (NR +
Is NH4+ an ionic compound?
Ammonium Sulfate is an ionic compound formed by two polyatomic ions, Ammonium NH+4 and Sulfate SO−4 . In order for these two polyatomic ions to bond the charges must be equal and opposite.
Is ammonium ion a cation?
The ammonium cation is a positively charged polyatomic ion with the chemical formula NH+4. It is formed by the protonation of ammonia (NH3).
Why is ammonium an ion?
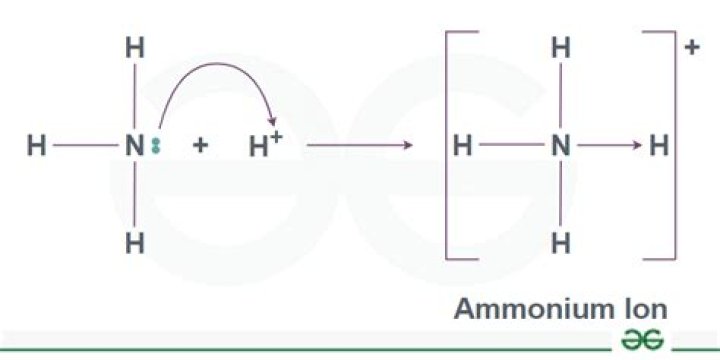
The lone electron pair on nitrogen also is available for making a covalent bond in which it supplies both of the electrons. An ammonia molecule can bind another proton (H+) and become an ammonium ion, NH4+, by the reaction shown on the right.
How many ions are in NH4+?
The dissociation is given below. One mole of ammonium phosphate dissolves to form 3 ammonium and one phosphate ion. Hence total of 4 ions is formed when ammonium phosphate dissolves in water.
Why is NH4 an ion?
Ammonium ions, NH4+, are formed by the transfer of a hydrogen ion from the hydrogen chloride to the lone pair of electrons on the ammonia molecule. The hydrogen’s electron is left behind on the chlorine to form a negative chloride ion.
How many atoms are in ammonium ion?
In an ammonium ion (NH+4) ion, there is one nitrogen atom and four hydrogen atoms.
Is ammonium ion ionic or covalent?
The ammonium ion, NH+4 , would have covalent bonds because both nitrogen and hydrogen are nonmetals. Of these covalent bonds, 1 of them would be considered a dative covalent bond, which is a covalent bond where both shared electrons come from the same atom.
Is ammonium molecular or ionic?
Ammonium ion is not a molecular ion because it is formed by the coordinate covalent bond between ammonia (NH3) and hydrogen ion (H+) .
What is the reaction between ammonium ion and NH3?
Ammonium ions in the soil solution enter into an equilibrium reaction with NH 3 in the soil solution. The soil solution NH 3 is, in turn, subject to gaseous loss to the atmosphere.
What is the formula for the ionic compound ammonium ion?
Ammonium Ion: Formula, Properties and Uses. He Ion ammonia Is a positively charged polyatomic cation whose chemical formula is NH 4 + . The molecule is not flat, but has the shape of a tetrahedron. The positively charged ammonium ion NH 4 + , The four hydrogen atoms make up the four corners of the tetrahedron.
What is the conversion of ammonia to ammonium?
In the ammonia step, the ammonia is converted by ammoniating bacteria into the ammonium ion (NH4 +). In the next step the ammonium is converted by nitrifying bacteria into nitrate (nitrification). This highly mobile form of nitrogen is most commonly absorbed by plant roots as well as by microorganisms in the soil.
Which of the following is an example of an ammonium ion?
An example of a reaction forming an ammonium ion is that between dimethylamine, (CH 3) 2NH, and an acid to give the dimethylammonium cation, (CH 3) 2NH +. 2: Quaternary ammonium cations have four organic groups attached to the nitrogen atom, they lack a hydrogen atom bonded to the nitrogen atom.



