What is a near equilibrium reaction?
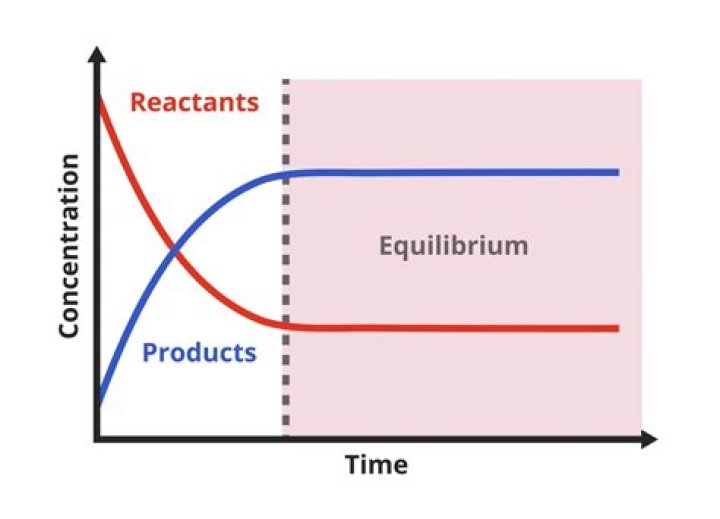
With a near equilibrium reaction, the rates of forward and reverse components are much greater than the overall net rate Delta G. Free Energy Change Close to zero. The rate of forward and reverse reaction are equal than most metabolic reactions are near equilibrium.
Is glycolysis at equilibrium?
For some of the enzymes in a pathway such as glycolysis, the reaction is essentially at equilibrium within the cell; the activity of such an enzyme is sufficiently high that the substrate is converted to product as fast as the substrate is supplied. Other cellular reactions are far from equilibrium.
How many carbons are there at the beginning of glycolysis?
Glycolysis begins with the six carbon ring-shaped structure of a single glucose molecule and ends with two molecules of a three-carbon sugar called pyruvate (Figure 1). Figure 1. Reactants and products of glycolysis. Glycolysis consists of ten steps divided into two distinct halves.
What is glycolysis reaction?
Abstract. Glycolysis is a linear metabolic pathway of enzyme-catalyzed reactions that converts glucose into two molecules of pyruvate in the presence of oxygen or two molecules of lactate in the absence of oxygen.
What is a near equilibrium enzyme?
With a near-equilibrium reaction, the rates of the forward and the reverse components of the reaction are much greater than the overall net rate and are similar to one another, for example: Example 2.
Why do enzymes catalyze near equilibrium reactions?
The former reactions are at or near equilibrium. What is this? Because enzymes can operate in either direction, relatively small changes in substrate concentration can change the net flow of substrates forward or backward through these reactions. Such reactions are said to be reversible.
Does glycolysis produce co2?
Glucose ( 6 carbon atoms) is split into 2 molecules of pyruvic acid (3 carbons each). This produces 2 ATP and 2 NADH. Glycolysis takes place in the cytoplasm. This breaks down the pyruvic acid to carbon dioxide.
How many carbons are lost during the process of glycolysis?
Overall, glycolysis converts one six-carbon molecule of glucose into two three-carbon molecules of pyruvate.
Does glycolysis produce CO2?
Which of the 10 reactions of glycolysis are?
Glycolysis Explained in 10 Easy Steps
- Step 1: Hexokinase.
- Step 2: Phosphoglucose Isomerase.
- Step 3: Phosphofructokinase.
- Step 4: Aldolase.
- Step 5: Triosephosphate isomerase.
- Step 6: Glyceraldehyde-3-phosphate Dehydrogenase.
- Step 7: Phosphoglycerate Kinase.
- Step 8: Phosphoglycerate Mutase.
What is a near-equilibrium reaction?
Most metabolic reactions are near-equilibrium reaction with ΔG = 0 (or close to it). This is an important concept in biochemistry.
What is the free energy of glycolysis at equilibrium?
Glycolysis Regulation. In the figure, the standard free energies and the concentrations were used to compute the total free energy differences between products and reactants at each step. Reactions at equilibrium have a free energy change of zero.
What is the chemical equation for glycolysis?
The Glycolytic Pathway (Embden-Meyerhof-Parnas Pathway) Glycolysis converts one C6 unit (glucose) to two C3 units (pyruvate) of lower energy in a process that harnesses the released free energy to synthesize ATP from ADP and Pi. Overall reaction – Glucose + 2NAD+ + 2ATP + 2P. i → 2NADH + 2pyruvate + 2ATP + 2H2O + 4H.
What is the overall reaction of the glycolytic pathway?
The Glycolytic Pathway (Embden-Meyerhof-Parnas Pathway) Glycolysis converts one C6 unit (glucose) to two C3 units (pyruvate) of lower energy in a process that harnesses the released free energy to synthesize ATP from ADP and Pi. Overall reaction – Glucose + 2NAD+ + 2ATP + 2P.



