What is a precipitate GCSE chemistry?
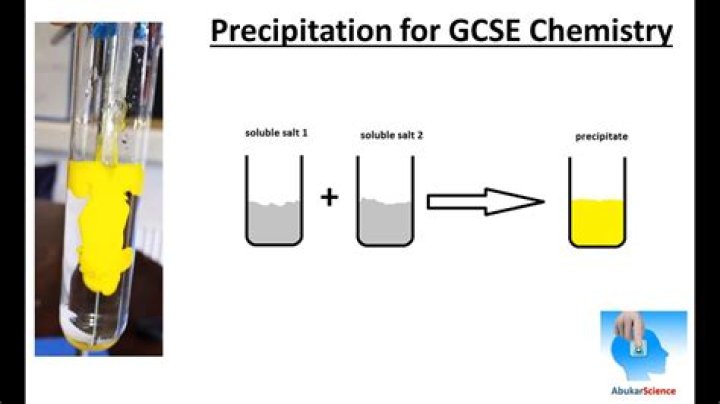
A precipitate is an insoluble product that forms when two solutions are mixed and react together. The reaction that produces a precipitate is called a precipitation reaction .
What is a precipitate as defined in chemistry?
A precipitate is an insoluble solid that emerges from a liquid solution. The process of making precipitate is called precipitation. Often the precipitate emerges as a suspension.
How do you know if it is a precipitate?
The solid is called a precipitate. Precipitation reactions occur when the cations of one reactant and the anions of a second reactant found in aqueous solutions combine to form an insoluble ionic solid that we call a precipitate. A precipitate forms if the product of the reaction of the ions is insoluble in water.
What is the precipitate test?
When metal ions combine with the hydroxide ions (OH -) from either sodium hydroxide solution or ammonia solution, they form insoluble precipitates with characteristic colours. Method: dissolve a small quantity of the substance in water. place about 5cm 3 of the solution into a test tube.
What is an example of precipitate in chemistry?
One of the best examples of precipitation reactions is the chemical reaction between potassium chloride and silver nitrate, in which solid silver chloride is precipitated out. In the above reaction, a white precipitate called silver chloride or AgCl is formed which is in the solid-state.
How do you find the precipitate in chemistry?
If the rules state that an ion is soluble, then it remains in its aqueous ion form. If an ion is insoluble based on the solubility rules, then it forms a solid with an ion from the other reactant. If all the ions in a reaction are shown to be soluble, then no precipitation reaction occurs.
What is the meaning of precipitate in chemistry?
Key Takeaways: Precipitate Definition in Chemistry. In chemistry, precipitate is both a verb and a noun. To precipitate is to form an insoluble compound, either by decreasing the solubility of a compound or by reacting two salt solutions. The solid that forms via a precipitation reaction is called the precipitate.
What is the chemical equation for the precipitation reaction?
This is the insoluble salt formed as a product of the precipitation reaction. The chemical equation for this precipitation reaction is provided below. AgNO3(aqueous) + KCl (aqueous) —–AgCl (precipitate) + KNO3(aqueous) In the above reaction, a white precipitate called silver chloride or AgCl is formed which is in the solid-state.
How do you test for precipitate formation in chemistry?
place about 5cm3 of the solution into a test tube. add a few drops of sodium hydroxide solution. record the colour of any precipitate that’s formed. add dilute sodium hydroxide solution until it is in excess and record the result.
What are the insoluble salts formed in precipitation reactions?
These insoluble salts formed in precipitation reactions are called precipitates. Precipitation reactions are usually double displacement reactions involving the production of a solid form residue called the precipitate.



