What is a receptor binding assay?
Receptor-binding assays have traditionally been a mainstay of drug development. These assays employ a simple biological preparation of cell membranes and a labeled compound to bind specific protein receptors in those membranes.
What is the difference between allosteric and orthosteric regulation?
Currently, there are two types of drugs on the market: orthosteric, which bind at the active site; and allosteric, which bind elsewhere on the protein surface, and allosterically change the conformation of the protein binding site.
How do you test for allosteric modulators?
A common method for screening for positive allosteric modulators is to examine a concentration-response (C/R) curve to the putative modulator in the presence of a single, low concentration of agonist.
What is the orthosteric binding site?
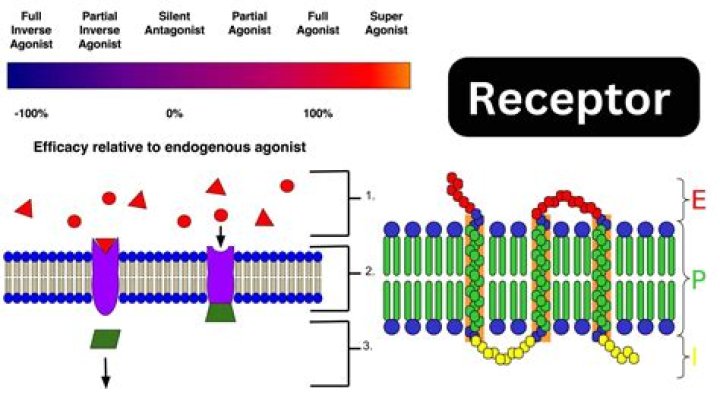
Box 2 | Endogenous allosteric modulators By definition, the orthosteric binding site on a receptor comprises amino acids that form contacts with the endogenous agonist for that receptor; this site has therefore specifically evolved to interact with an endogenous hormone or neurotransmitter.
How do you determine binding affinity experimentally?
Binding affinity is typically measured and reported by the equilibrium dissociation constant (KD), which is used to evaluate and rank order strengths of bimolecular interactions. The smaller the KD value, the greater the binding affinity of the ligand for its target.
What are orthosteric inhibitors?
Notes: An orthosteric inhibitor (represented as a purple rectangle) interferes directly with the protein-protein interface, disrupting binding, whilst an allosteric inhibitor (represented as a green triangle) induces a conformational change to the binding interface region of the protein that indirectly disables binding …
What is an orthosteric modulator?
The site that an allosteric modulator binds to (i.e., an allosteric site) is not the same one to which an endogenous agonist of the receptor would bind (i.e., an orthosteric site). Modulators and agonists can both be called receptor ligands.
What does Orthosteric mean?
Filters. (biochemistry) Describing the primary, unmodulated binding site (on a receptor) of a ligand. adjective.
What is Orthosteric inhibitor?
How many types of assay are there?
Assays can be divided into three main categories based on the type of sample used – ligand-binding assays that measure binding between a ligand and a receptor, immunoassays that detect antibody-antigen binding, and bioassays that measure biological activity in response to certain stimuli.
Do allosteric modulators interact with orthosteric receptor binding sites?
However, recent developments have highlighted the fact that drugs can interact with binding sites on the receptor molecule that are distinct from the orthosteric site, known as allosteric sites. Allosteric modulators could offer several advantages over orthosteric ligands, including greater selectivity and saturability of their effect.
What is a radioactively labeled ligand binding assay?
Radioactively labeled ligand binding assay is applied to detect its binding to a target. The most popular assay for membrane-bound targets. Good robustness, precise determination of receptor density and distribution, ligand binding sites and affinity. High cost and hazards of handling high levels of radioactivity.
What is standard orthosteric drug–receptor theory?
Standard orthosteric drug–receptor theory, which forms the basis for the quantitative comparison of the drug affinity values that are derived in most drug discovery programmes, is based on the law of mass action and some simplifying assumptions 5.
Are there any compounds that have no effect on orthosteric ligands?
The converse situation is also true; there are compounds that can exert a profound effect on the binding of some orthosteric ligands and have no effect on others, all at the same receptor.



