What is diffusion controlled process?
Diffusion-controlled (or diffusion-limited) reactions are reactions in which the reaction rate is equal to the rate of transport of the reactants through the reaction medium (usually a solution). Examples are those involving catalysis and enzymatic reactions.
What are the two types of quenching?
There are two different ways of quenching: static and dynamic quenching.
What is collision quenching?
Collisional quenching occurs when the excited fluorophore experiences contact with an atom or molecule that can facilitate non-radiative transitions to the ground state. Excited-state molecule collides with quencher molecule and returns to ground state non-radiatively.
What is diffusion-controlled enzyme?
A diffusion-limited enzyme catalyses a reaction so efficiently that the rate limiting step is that of substrate diffusion into the active site, or product diffusion out. Most enzymes catalyse their reactions to a rate that is 1,000-10,000 times slower than this limit.
What are types of quenching?
There are ten quenching methods in the heat treatment process, which are:
- single-medium (water, oil, air) quenching;
- interrupted quenching;
- martempering;
- martempering below MS point;
- isothermal quenching of bainite;
- compound quenching;
- precooled isothermal quenching;
- delayed cooling quenching;
What is the function of quenching?
In materials science, quenching is the rapid cooling of a workpiece in water, oil or air to obtain certain material properties. A type of heat treating, quenching prevents undesired low-temperature processes, such as phase transformations, from occurring.
What is quenching heat treatment?
Quenching is when a part that has been heated to a given metal transformation temperature is cooled quickly. Compared to slow cooling, rapid cooling modifies the metal’s structure and thereby its hardness characteristics (surface or core) and elasticity.
How does quenching take place?
Dynamic quenching is caused by interaction between two light-sensitive molecules; a donor and acceptor. The donor fluorophore transfers energy to the acceptor, which may then emit light itself or completely absorb the energy. In dynamic quenching, electron excitation takes place before the quenching process.
What is the purpose of quenching?
Quenching improves a metal’s performance by rapidly cooling the heated metal, thereby altering its molecular structure and increasing its hardness. The rate of quenching may be adjusted to achieve the desired properties.
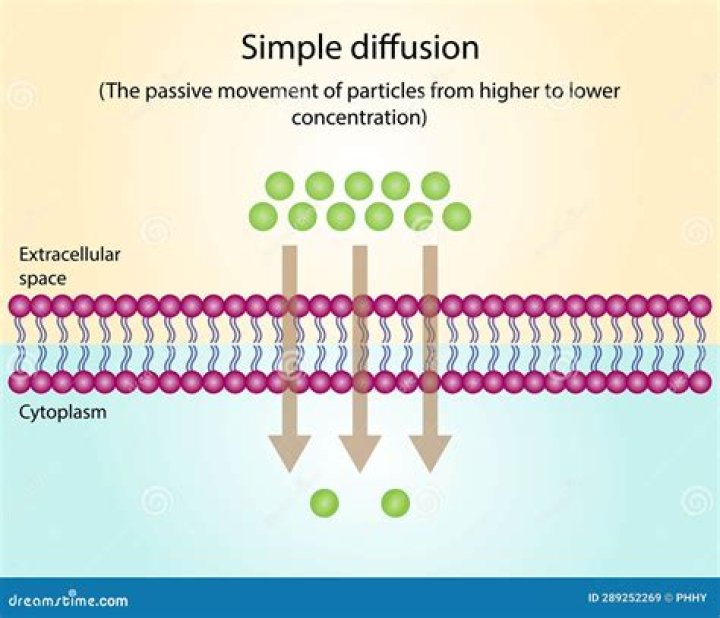
What is meant by diffusion control reaction?
Diffusion-controlled reaction. Diffusion control is more likely in solution where diffusion of reactants is slower due to the greater number of collisions with solvent molecules. Reactions where the activated complex forms easily and the products form rapidly are most likely to be limited by diffusion control.
What are the advantages of quenching a material?
In some cases, quenching can make a material more ductile, and softer. In the case of most steels, you saw that the speed of quenching allowed you to get a metastable martensite, instead of the thermodynamically favored ferrite. However, in other materials (or some unique steels), quenching can let you avoid a strengthening or embrittling phase.
What are the different types of quenching media?
The most common quenching media are water, brine (salt water), oil, liquid nitrogen, and air. Each of these media has different advantages and disadvantages.
What is the relationship between quencher concentration and aggregation number?
In the static case, the estimated aggregation number will decrease with quencher concentration from the weight average aggregation number in the limit of zero quencher concentration. In the dynamic case, the number average aggregation number is obtained, independent of quencher concentration.



