What is integration in proton NMR?
The integration in NMR tells us the number of protons represented by a given signal. To be more accurate, let’s mention that it is the ratio of the protons behind each signal.
How are integrations determined in a proton NMR experiment?
When the integrator trace crosses a peak or group of peaks, it gains height. The height gained is proportional to the area under the peak or group of peaks. You measure the height gained at each peak or group of peaks by measuring the distances shown in green in the diagram above – and then find their ratio.
Can you integrate NMR?
Signal integration The computer in an NMR instrument can be instructed to automatically integrate the area under a signal or group of signals. This is very useful, because in 1H-NMR spectroscopy the area under a signal is proportional to the number of hydrogens to which the peak corresponds.
Does 13c NMR have integration?
Integration is almost useless in a regular 13C NMR spectrum because of uneven nuclear Overhauser effect (NOE) enhancement of the signals by decoupling and long longitudinal relaxation times (T1’s).
How do you find integration?
Basic Integration Formulas
- ∫ xn.dx = x(n + 1)/(n + 1)+ C.
- ∫ 1.dx = x + C.
- ∫ ex.dx = ex + C.
- ∫1/x.dx = log|x| + C.
- ∫ ax.dx = ax /loga+ C.
- ∫ ex[f(x) + f'(x)].dx = ex.f(x) + C.
How do you calculate integration?
What is the integration ratio in NMR?
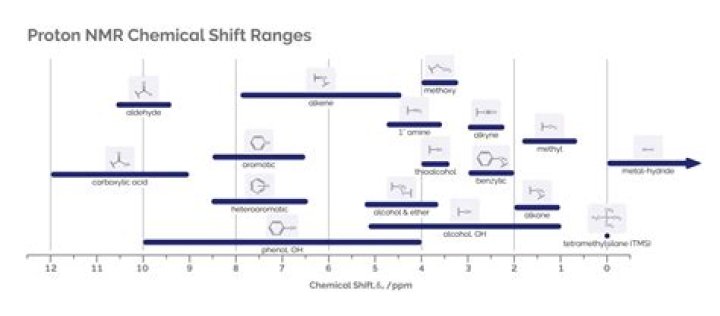
Chemical shift can show how many different types of hydrogens are found in a molecule; integration reveals the number of hydrogens of each type. An integrator trace (or integration trace) can be used to find the ratio of the numbers of hydrogen atoms in different environments in an organic compound.
What is proton-decoupled 13C NMR?
The splitting of a resonance for a 13C atom by hydrogen can be eliminated to generate a singlet by a technique called proton decoupling. The resulting spectrum is called a proton-decoupled NMR spectrum. This combination of field and radio frequency causes changes in spin states of hydrogen nuclei.
Which molecules will have 4 peaks in its 13C NMR spectrum?
group. However, in propanal, all the carbons are in completely different environments, and the spectrum will have three peaks. There are four alcohols with the molecular formula C4H10O….Working out Structures from C-13 NMR Spectra.
| carbon environment | chemical shift (ppm) |
|---|---|
| C=O | 150 – 200 |
What is an integrator trace in NMR?
An integrator trace is a computer generated line which is superimposed on a proton NMR spectra. In the diagram, the integrator trace is shown in red. An integrator trace measures the relative areas under the various peaks in the spectrum.
Is vanillin an anti-mutation agent?
/ALTERNATIVE and IN VITRO TESTS/ Vanillin is responsible for the flavor and smell of vanilla, a widely used flavoring agent. Previous studies showed that vanillin could enhance the repair of mutations and thus function as an anti-mutagen.
What is the molecular formula of vanillin?
Vanillin PubChem CID 1183 Structure Find Similar Structures Chemical Safety Laboratory Chemical Safety Summary (LCSS Molecular Formula C8H8O3 Synonyms vanillin 4-Hydroxy-3-methoxybenzaldehyde
What is the function of vanillin?
It has a role as a plant metabolite, a flavouring agent, an antioxidant and an anticonvulsant. It is a member of phenols, a monomethoxybenzene and a member of benzaldehydes. Vanillin appears as white or very slightly yellow needles. Vanillin is the primary component of the extract of the vanilla bean.



